Clinical quantitative susceptibility mapping (QSM): Biometal imaging and its emerging roles in patient care
- PMID: 28295954
- PMCID: PMC5592126
- DOI: 10.1002/jmri.25693
Clinical quantitative susceptibility mapping (QSM): Biometal imaging and its emerging roles in patient care
Abstract
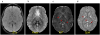
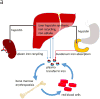
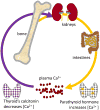
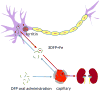
Quantitative susceptibility mapping (QSM) has enabled magnetic resonance imaging (MRI) of tissue magnetic susceptibility to advance from simple qualitative detection of hypointense blooming artifacts to precise quantitative measurement of spatial biodistributions. QSM technology may be regarded to be sufficiently developed and validated to warrant wide dissemination for clinical applications of imaging isotropic susceptibility, which is dominated by metals in tissue, including iron and calcium. These biometals are highly regulated as vital participants in normal cellular biochemistry, and their dysregulations are manifested in a variety of pathologic processes. Therefore, QSM can be used to assess important tissue functions and disease. To facilitate QSM clinical translation, this review aims to organize pertinent information for implementing a robust automated QSM technique in routine MRI practice and to summarize available knowledge on diseases for which QSM can be used to improve patient care. In brief, QSM can be generated with postprocessing whenever gradient echo MRI is performed. QSM can be useful for diseases that involve neurodegeneration, inflammation, hemorrhage, abnormal oxygen consumption, substantial alterations in highly paramagnetic cellular iron, bone mineralization, or pathologic calcification; and for all disorders in which MRI diagnosis or surveillance requires contrast agent injection. Clinicians may consider integrating QSM into their routine imaging practices by including gradient echo sequences in all relevant MRI protocols.
Level of evidence: 1 Technical Efficacy: Stage 5 J. Magn. Reson. Imaging 2017;46:951-971.
Keywords: biometals; quantitative susceptibility mapping.
© 2017 International Society for Magnetic Resonance in Medicine.
Figures





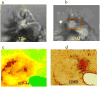
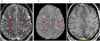
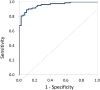
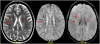
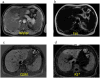
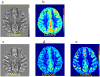
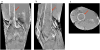
References
-
- de Rochefort L, Liu T, Kressler B, et al. Quantitative susceptibility map reconstruction from MR phase data using bayesian regularization: validation and application to brain imaging. Magn Reson Med. 2010;63(1):194–206. - PubMed
-
- Reichenbach JR, Schweser F, Serres B, Deistung A. Quantitative Susceptibility Mapping: Concepts and Applications. Clin Neuroradiol. 2015;25(2):225–230. - PubMed
-
- Haacke EM, Liu S, Buch S, Zheng W, Wu D, Ye Y. Quantitative susceptibility mapping: current status and future directions. Magnetic resonance imaging. 2015;33(1):1–25. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

