Interstrain differences in the expression and activity of Cyp2a5 in the mouse liver
- PMID: 28298240
- PMCID: PMC5353797
- DOI: 10.1186/s13104-017-2435-x
Interstrain differences in the expression and activity of Cyp2a5 in the mouse liver
Abstract
Background: Cytochrome P450 2A5 (Cyp2a5), a mouse enzyme orthologous of human CYP2A6, catalyzes a number of toxicologically important reactions, including the metabolism of nicotine, aflatoxin B1, and several other xeno- and endobiotics. Cyp2a5 expression is complex and not yet fully understood. We investigated inter-strain differences in the activity and mRNA expression of hepatic Cyp2a5. Cyp1a1/2 and Cyp2b9/10 activities were evaluated for comparative purposes. Data on the interstrain differences in the expression and activity of Cyp2a5 are important to select a suitable mouse model for studying CYP2A6-mediated metabolism.
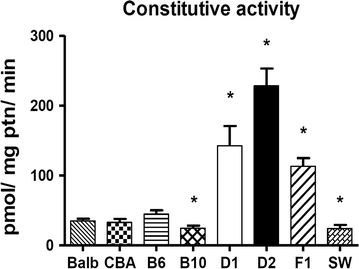
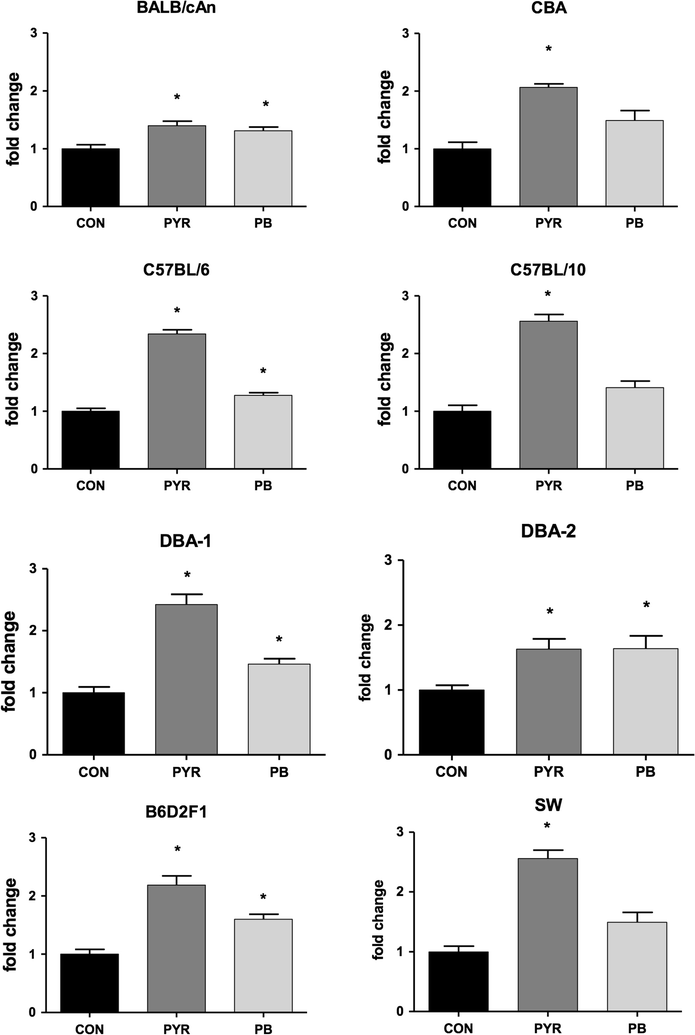
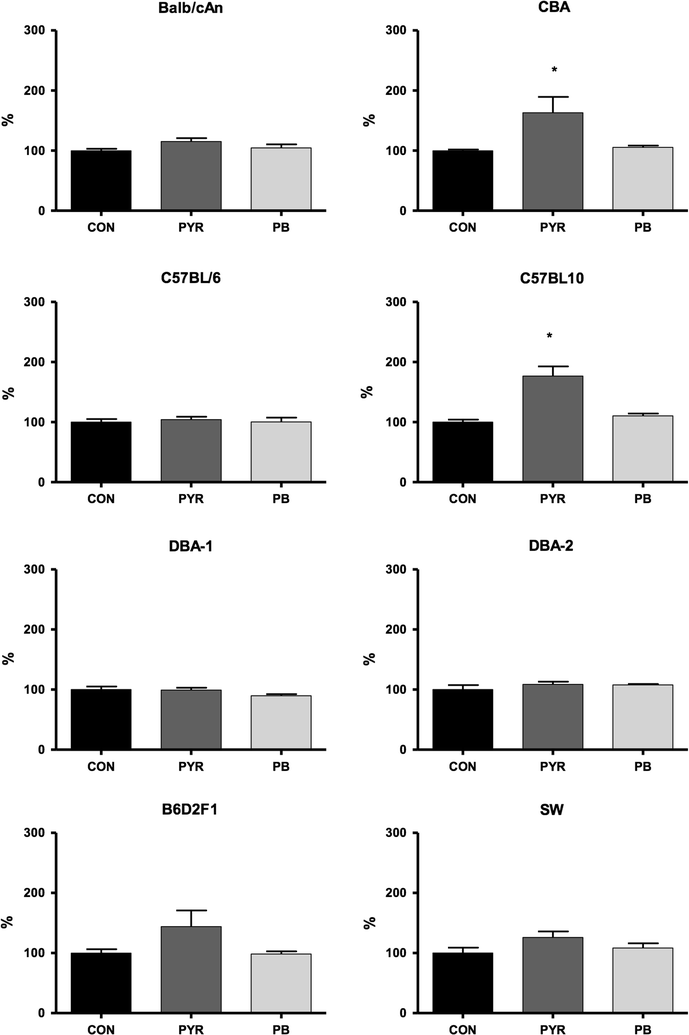
Results: Activity of Cyp2a5 (coumarin 7-hydroxylase) was highest in DBA-2 and DBA-1, intermediate in B6D2F1 (hybrid) and low in the remaining strains (C57BL/6, C57BL/10, CBA, BALB/cAn, SW). Contrasting with the activity, background levels of Cyp2a4/5 mRNA did not differ between high- and low-activity murine strains. Phenobarbital (PB, 80 mg/kg body weight/day × 3 days, i.p.) increased Cyp2a5, Cyp1a1/2 (ethoxyresorufin-O-deethylase) and Cyp2b9/10 (bezyloxyresorufin-O-debenzylase) activities while only Cyp2a5 was enhanced by pyrazole (PYR, 100 mg/kg body weight/day × 3 days, i.p.). Inductions of Cyp2a5 activity by PYR and PB were accompanied by increases of Cyp2a4/5 mRNA. PYR and PB did not upregulate heme oxygenase-1 (hmox-1) mRNA expression in any strain, a finding that is apparently at odds with the notion that Cyp2a5 and hmox-1 inductions are coordinated events.
Conclusions: Since background levels of Cyp2a4/5 gene transcripts of high-activity strains did not differ from those of low-activity mice, distinct constitutive activities did not result from different transcription rates and/or mRNA half-lives. Results therefore suggested that interstrain differences in constitutive activity of Cyp2a5 possibly arise from distinct translation efficiencies, protein half-lives and/or enzyme kinetics toward the substrate. Data from this study indicated that all tested strains are suitable models for studying toxicants that are substrates for human CYP2A6; DBA-2, DBA-1 and the hybrid B62DF1, however, have the advantage of presenting high constitutive activities of Cyp2a5.
Keywords: Coumarin 7-hydroxylase; Cyp2a4; Cyp2a5; Heme oxygenase; Liver toxicity.
Figures


Similar articles
-
Circadian expression of the steroid 15 alpha-hydroxylase (Cyp2a4) and coumarin 7-hydroxylase (Cyp2a5) genes in mouse liver is regulated by the PAR leucine zipper transcription factor DBP.Mol Cell Biol. 1999 Oct;19(10):6488-99. doi: 10.1128/MCB.19.10.6488. Mol Cell Biol. 1999. PMID: 10490589 Free PMC article.
-
Induction of CYP2A5 by pyrazole and its derivatives in mouse primary hepatocytes.Arch Toxicol. 1998 May;72(6):336-41. doi: 10.1007/s002040050511. Arch Toxicol. 1998. PMID: 9657280
-
Evidence for induced microsomal bilirubin degradation by cytochrome P450 2A5.Biochem Pharmacol. 2005 Nov 15;70(10):1527-35. doi: 10.1016/j.bcp.2005.08.009. Epub 2005 Sep 23. Biochem Pharmacol. 2005. PMID: 16183037
-
Function and regulation of the Cyp2a5/CYP2A6 genes in response to toxic insults in the liver.Curr Drug Metab. 2013 Jan;14(1):137-50. Curr Drug Metab. 2013. PMID: 22497566 Review.
-
CYP2A5 induction and hepatocellular stress: an adaptive response to perturbations of heme homeostasis.Curr Drug Metab. 2011 Feb;12(2):186-97. doi: 10.2174/138920011795016845. Curr Drug Metab. 2011. PMID: 21395539 Review.
Cited by
-
Maternal smoking and its short- or long-term impact on offspring liver pathologies: a review of experimental and clinical studies.Toxicol Res. 2024 Dec 21;41(2):123-129. doi: 10.1007/s43188-024-00271-y. eCollection 2025 Mar. Toxicol Res. 2024. PMID: 40013082 Review.
-
Suppression of NNK Metabolism by Anthocyanin-Rich Haskap Berry Supplementation Through Modulation of P450 Enzymes.Pharmaceuticals (Basel). 2024 Nov 30;17(12):1615. doi: 10.3390/ph17121615. Pharmaceuticals (Basel). 2024. PMID: 39770457 Free PMC article.
-
Strain-specific altered nicotine metabolism in 3,3'-diindolylmethane (DIM) exposed mice.Biopharm Drug Dispos. 2019 May;40(5-6):188-194. doi: 10.1002/bdd.2182. Epub 2019 May 20. Biopharm Drug Dispos. 2019. PMID: 31016737 Free PMC article.
-
Mouse strain-specific acute respiratory effects of nicotine unrelated to nicotine metabolism.Toxicol Mech Methods. 2019 Sep;29(7):542-548. doi: 10.1080/15376516.2019.1628141. Epub 2019 Jun 21. Toxicol Mech Methods. 2019. PMID: 31172850 Free PMC article.
-
Increased toxicity and retention of perflourooctane sulfonate (PFOS) in humanized CYP2B6-Transgenic mice compared to Cyp2b-null mice is relieved by a high-fat diet (HFD).Food Chem Toxicol. 2021 Jun;152:112175. doi: 10.1016/j.fct.2021.112175. Epub 2021 Apr 8. Food Chem Toxicol. 2021. PMID: 33838175 Free PMC article.
References
-
- Chomarat P, Sipowicz MA, Diwan BA, Fornwald LW, Awasthi YC, Anver MR, Rice JM, Anderson LM, Wild CP. Distinct time courses of increase in cytochromes P450 1A2, 2A5 and glutathione S-transferases during the progressive hepatitis associated with Helicobacter hepaticus. Carcinogenesis. 1997;18(11):2179–2190. doi: 10.1093/carcin/18.11.2179. - DOI - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

