The short variant of the mitochondrial dynamin OPA1 maintains mitochondrial energetics and cristae structure
- PMID: 28298442
- PMCID: PMC5409478
- DOI: 10.1074/jbc.M116.762567
The short variant of the mitochondrial dynamin OPA1 maintains mitochondrial energetics and cristae structure
Abstract
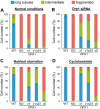
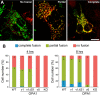
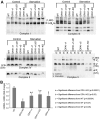
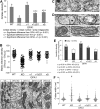
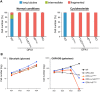
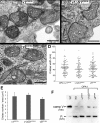
The protein optic atrophy 1 (OPA1) is a dynamin-related protein associated with the inner mitochondrial membrane and functions in mitochondrial inner membrane fusion and cristae maintenance. Inner membrane-anchored long OPA1 (L-OPA1) undergoes proteolytic cleavage resulting in short OPA1 (S-OPA1). It is often thought that S-OPA1 is a functionally insignificant proteolytic product of L-OPA1 because the accumulation of S-OPA1 due to L-OPA1 cleavage is observed in mitochondrial fragmentation and dysfunction. However, cells contain a mixture of both L- and S-OPA1 in normal conditions, suggesting the functional significance of maintaining both OPA1 forms, but the differential roles of L- and S-OPA1 in mitochondrial fusion and energetics are ill-defined. Here, we examined mitochondrial fusion and energetic activities in cells possessing L-OPA1 alone, S-OPA1 alone, or both L- and S-OPA1. Using a mitochondrial fusion assay, we established that L-OPA1 confers fusion competence, whereas S-OPA1 does not. Remarkably, we found that S-OPA1 alone without L-OPA1 can maintain oxidative phosphorylation function as judged by growth in oxidative phosphorylation-requiring media, respiration measurements, and levels of the respiratory complexes. Most strikingly, S-OPA1 alone maintained normal mitochondrial cristae structure, which has been commonly assumed to be the function of OPA1 oligomers containing both L- and S-OPA1. Furthermore, we found that the GTPase activity of OPA1 is critical for maintaining cristae tightness and thus energetic competency. Our results demonstrate that, contrary to conventional notion, S-OPA1 is fully competent for maintaining mitochondrial energetics and cristae structure.
Keywords: OPA1; bioenergetics; cristae; mitochondria; mitochondrial disease; mitochondrial fusion; mitochondrial metabolism; mitochondrial respiratory chain complex.
© 2017 by The American Society for Biochemistry and Molecular Biology, Inc.
Conflict of interest statement
The authors declare that they have no conflicts of interest with the contents of this article
Figures




Similar articles
-
The short variant of optic atrophy 1 (OPA1) improves cell survival under oxidative stress.J Biol Chem. 2020 May 8;295(19):6543-6560. doi: 10.1074/jbc.RA119.010983. Epub 2020 Apr 3. J Biol Chem. 2020. PMID: 32245890 Free PMC article.
-
Eight human OPA1 isoforms, long and short: What are they for?Biochim Biophys Acta Bioenerg. 2018 Apr;1859(4):263-269. doi: 10.1016/j.bbabio.2018.01.005. Epub 2018 Jan 31. Biochim Biophys Acta Bioenerg. 2018. PMID: 29382469 Review.
-
Identification of new OPA1 cleavage site reveals that short isoforms regulate mitochondrial fusion.Mol Biol Cell. 2021 Jan 15;32(2):157-168. doi: 10.1091/mbc.E20-09-0605. Epub 2020 Nov 25. Mol Biol Cell. 2021. PMID: 33237841 Free PMC article.
-
OPA1 controls apoptotic cristae remodeling independently from mitochondrial fusion.Cell. 2006 Jul 14;126(1):177-89. doi: 10.1016/j.cell.2006.06.025. Cell. 2006. PMID: 16839885
-
A cut short to death: Parl and Opa1 in the regulation of mitochondrial morphology and apoptosis.Cell Death Differ. 2007 Jul;14(7):1275-84. doi: 10.1038/sj.cdd.4402145. Epub 2007 Apr 20. Cell Death Differ. 2007. PMID: 17464328 Review.
Cited by
-
Mitochondrial OMA1 and OPA1 as Gatekeepers of Organellar Structure/Function and Cellular Stress Response.Front Cell Dev Biol. 2021 Mar 25;9:626117. doi: 10.3389/fcell.2021.626117. eCollection 2021. Front Cell Dev Biol. 2021. PMID: 33842456 Free PMC article. Review.
-
Melatonin Attenuates Cardiac Reperfusion Stress by Improving OPA1-Related Mitochondrial Fusion in a Yap-Hippo Pathway-Dependent Manner.J Cardiovasc Pharmacol. 2019 Jan;73(1):27-39. doi: 10.1097/FJC.0000000000000626. J Cardiovasc Pharmacol. 2019. PMID: 30418242 Free PMC article.
-
Mitochondrial fusion is required for regulation of mitochondrial DNA replication.PLoS Genet. 2019 Jun 6;15(6):e1008085. doi: 10.1371/journal.pgen.1008085. eCollection 2019 Jun. PLoS Genet. 2019. PMID: 31170154 Free PMC article.
-
Unveiling the potential of mitochondrial dynamics as a therapeutic strategy for acute kidney injury.Front Cell Dev Biol. 2023 Aug 11;11:1244313. doi: 10.3389/fcell.2023.1244313. eCollection 2023. Front Cell Dev Biol. 2023. PMID: 37635869 Free PMC article. Review.
-
The mycotoxin viriditoxin induces leukemia- and lymphoma-specific apoptosis by targeting mitochondrial metabolism.Cell Death Dis. 2022 Nov 8;13(11):938. doi: 10.1038/s41419-022-05356-w. Cell Death Dis. 2022. PMID: 36347842 Free PMC article.
References
-
- Koshiba T., Detmer S. A., Kaiser J. T., Chen H., McCaffery J. M., and Chan D. C. (2004) Structural basis of mitochondrial tethering by mitofusin complexes. Science 305, 858–862 - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

