Impact of Pressurized Intraperitoneal Aerosol Chemotherapy on Quality of Life and Symptoms in Patients with Peritoneal Carcinomatosis: A Retrospective Cohort Study
- PMID: 28316621
- PMCID: PMC5339484
- DOI: 10.1155/2017/4596176
Impact of Pressurized Intraperitoneal Aerosol Chemotherapy on Quality of Life and Symptoms in Patients with Peritoneal Carcinomatosis: A Retrospective Cohort Study
Abstract
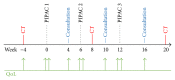
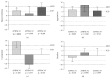
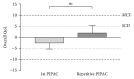
Background. Peritoneal cancer treatment aims to prolong survival, but preserving Quality of Life (QoL) under treatment is also a priority. Pressurized Intraperitoneal Aerosol Chemotherapy (PIPAC) is a novel minimally invasive repeatable treatment modality. The aim of the present study was to assess QoL in our cohort of PIPAC patients. Methods. Analysis of all consecutive patients included from the start of PIPAC program (January 2015). QoL (0-100: optimal) and symptoms (no symptom: 0-100) were measured prospectively before and after every PIPAC procedure using EORTC QLQ-C30. Results. Forty-two patients (M : F = 8 : 34, median age 66 (59-73) years) had 91 PIPAC procedures in total (1 : 4x, 17 : 3x, 12 : 2x, and 12 : 1x). Before first PIPAC, baseline QoL was measured as median of 66 ± 2.64. Prominent complaints were fatigue (32 ± 4.3) and digestive symptoms as diarrhea (17 ± 3.75), constipation (17 ± 4.13), and nausea (7 ± 2.54). Overall Quality of Life was 64 ± 3.75 after PIPAC#1 (p = 0.57), 61 ± 4.76 after PIPAC#2 (p = 0.89), and 70 ± 6.67 after PIPAC#3 (p = 0.58). Fatigue symptom score was 44 ± 4.86 after PIPAC#1 and 47 ± 6.69 and 34 ± 7.85 after second and third applications, respectively (p = 0.40). Diarrhea (p = 0.31), constipation (p = 0.76), and nausea (p = 0.66) did not change significantly under PIPAC treatment. Conclusion. PIPAC treatment of peritoneal carcinomatosis had no negative impact on patients' overall QoL and its components or on main symptoms. This study was registered online on Research Registry (UIN: 1608).
Conflict of interest statement
The authors declare that they have no competing interests.
Figures







Similar articles
-
Randomized control trial comparing quality of life of patients with end-stage peritoneal metastasis treated with pressurized intraperitoneal aerosol chemotherapy (PIPAC) and intravenous chemotherapy.Pleura Peritoneum. 2018 Sep 26;3(3):20180110. doi: 10.1515/pp-2018-0110. eCollection 2018 Sep 1. Pleura Peritoneum. 2018. PMID: 30911661 Free PMC article.
-
Quality of life of patients with end-stage peritoneal metastasis treated with Pressurized IntraPeritoneal Aerosol Chemotherapy (PIPAC).Eur J Surg Oncol. 2015 Oct;41(10):1379-85. doi: 10.1016/j.ejso.2015.06.001. Epub 2015 Jun 21. Eur J Surg Oncol. 2015. PMID: 26138283
-
Feasibility and Safety of Pressurized Intraperitoneal Aerosol Chemotherapy for Peritoneal Carcinomatosis: A Retrospective Cohort Study.Gastroenterol Res Pract. 2017;2017:6852749. doi: 10.1155/2017/6852749. Epub 2017 Feb 26. Gastroenterol Res Pract. 2017. PMID: 28331493 Free PMC article.
-
A systematic review on quality of life (QoL) of patients with peritoneal metastasis (PM) who underwent pressurized intraperitoneal aerosol chemotherapy (PIPAC).Pleura Peritoneum. 2022 Apr 21;7(2):39-49. doi: 10.1515/pp-2021-0154. eCollection 2022 Jun 1. Pleura Peritoneum. 2022. PMID: 35812010 Free PMC article. Review.
-
Systematic review of patient reported outcomes (PROs) and quality of life measures after pressurized intraperitoneal aerosol chemotherapy (PIPAC).Surg Oncol. 2020 Dec;35:97-105. doi: 10.1016/j.suronc.2020.08.012. Epub 2020 Aug 20. Surg Oncol. 2020. PMID: 32862112
Cited by
-
Comparing patient reported abdominal pain between patients treated with oxaliplatin-based pressurized intraperitoneal aerosol chemotherapy (PIPAC-OX) and primary colorectal cancer surgery.Sci Rep. 2023 Nov 22;13(1):20458. doi: 10.1038/s41598-023-47510-0. Sci Rep. 2023. PMID: 37993560 Free PMC article.
-
Pressurized intraperitoneal aerosol chemotherapy (PIPAC) might increase the risk of anastomotic leakage compared to HIPEC: an experimental study.Surg Endosc. 2020 Jul;34(7):2939-2946. doi: 10.1007/s00464-019-07076-3. Epub 2019 Aug 27. Surg Endosc. 2020. PMID: 31456025
-
Treatment Response After Pressurized IntraPeritoneal Aerosol Chemotherapy (PIPAC) for Peritoneal Metastases of Colorectal Originf.Ann Surg Open. 2022 Oct 24;3(4):e203. doi: 10.1097/AS9.0000000000000203. eCollection 2022 Dec. Ann Surg Open. 2022. PMID: 37600288 Free PMC article.
-
Pressurized intraperitoneal aerosol chemotherapy: a review of the introduction of a new surgical technology using the IDEAL framework.BJS Open. 2020 Apr;4(2):206-215. doi: 10.1002/bjs5.50257. Epub 2020 Jan 19. BJS Open. 2020. PMID: 31957257 Free PMC article. Review.
-
Feasibility, Safety, and Efficacy of Pressurized Intraperitoneal Aerosol Chemotherapy (PIPAC) for Peritoneal Metastasis: A Registry Study.Gastroenterol Res Pract. 2018 Oct 24;2018:2743985. doi: 10.1155/2018/2743985. eCollection 2018. Gastroenterol Res Pract. 2018. PMID: 30473706 Free PMC article.
References
-
- Lemmens V. E., Klaver Y. L., Verwaal V. J., Rutten H. J., Coebergh J. W. W., De Hingh I. H. Predictors and survival of synchronous peritoneal carcinomatosis of colorectal origin: A population-based study. International Journal of Cancer. 2011;128(11):2717–2725. doi: 10.1002/ijc.25596. - DOI - PubMed
-
- Klaver Y. L. B., Lemmens V. E. P. P., Creemers G. J., Rutten H. J. T., Nienhuijs S. W., de Hingh I. H. J. T. Population-based survival of patients with peritoneal carcinomatosis from colorectal origin in the era of increasing use of palliative chemotherapy. Annals of Oncology. 2011;22(10):2250–2256. doi: 10.1093/annonc/mdq762. - DOI - PubMed
-
- Ceelen W. P., Påhlman L., Mahteme H. Pharmacodynamic aspects of intraperitoneal cytotoxic therapy. Cancer Treatment and Research. 2007;134:195–214. - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

