Soluble oligomeric amyloid-β induces calcium dyshomeostasis that precedes synapse loss in the living mouse brain
- PMID: 28327181
- PMCID: PMC5361864
- DOI: 10.1186/s13024-017-0169-9
Soluble oligomeric amyloid-β induces calcium dyshomeostasis that precedes synapse loss in the living mouse brain
Abstract
Background: Amyloid-β oligomers (oAβ) are thought to mediate neurotoxicity in Alzheimer's disease (AD), and previous studies in AD transgenic mice suggest that calcium dysregulation may contribute to these pathological effects. Even though AD mouse models remain a valuable resource to investigate amyloid neurotoxicity, the concomitant presence of soluble Aβ species, fibrillar Aβ, and fragments of amyloid precursor protein (APP) complicate the interpretation of the phenotypes.
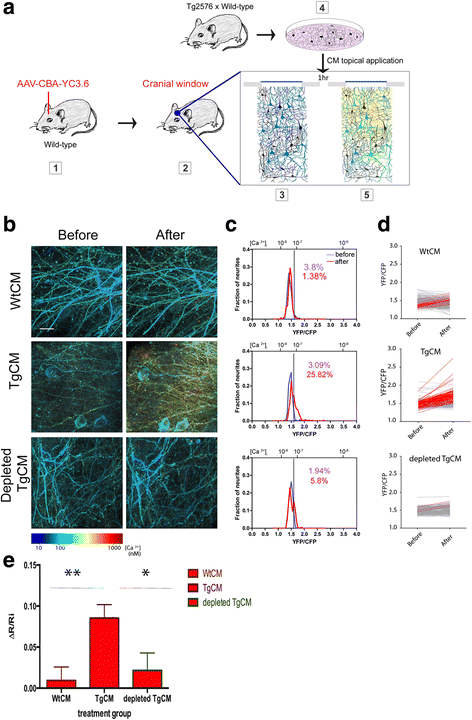
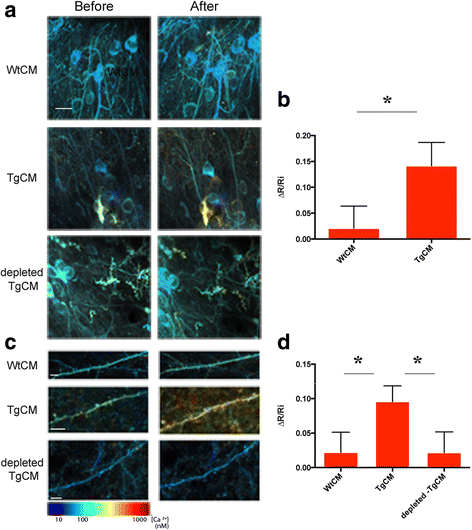
Method: To explore the specific contribution of soluble oligomeric Aβ (oAβ) to calcium dyshomeostasis and synaptic morphological changes, we acutely exposed the healthy mouse brain, at 3 to 6 months of age, to naturally occurring soluble oligomers and investigated their effect on calcium levels using in vivo multiphoton imaging.
Results: We observed a dramatic increase in the levels of neuronal resting calcium, which was dependent upon extracellular calcium influx and activation of NMDA receptors. Ryanodine receptors, previously implicated in AD models, did not appear to be primarily involved using this experimental setting. We used the high resolution cortical volumes acquired in-vivo to measure the effect on synaptic densities and observed that, while spine density remained stable within the first hour of oAβ exposure, a significant decrease in the number of dendritic spines was observed 24 h post treatment, despite restoration of intraneuronal calcium levels at this time point.
Conclusions: These observations demonstrate a specific effect of oAβ on NMDA-mediated calcium influx, which triggers synaptic collapse in vivo. Moreover, this work leverages a method to quantitatively measure calcium concentration at the level of neuronal processes, cell bodies and single synaptic elements repeatedly and thus can be applicable to testing putative drugs and/or other intervention methodologies.
Keywords: Alzheimer’s disease; Amyloid β oligomers; Calcium; In vivo imaging.
Figures



References
-
- Spires TL, Meyer-Luehmann M, Stern EA, McLean PJ, Skoch J, Nguyen PT, Bacskai BJ, Hyman BT. Dendritic spine abnormalities in amyloid precursor protein transgenic mice demonstrated by gene transfer and intravital multiphoton microscopy. J Neurosci. 2005;25:7278–7287. doi: 10.1523/JNEUROSCI.1879-05.2005. - DOI - PMC - PubMed
-
- Wu HY, Hudry E, Hashimoto T, Kuchibhotla K, Rozkalne A, Fan Z, Spires-Jones T, Xie H, Arbel-Ornath M, Grosskreutz CL, et al. Amyloid beta induces the morphological neurodegenerative triad of spine loss, dendritic simplification, and neuritic dystrophies through calcineurin activation. J Neurosci. 2010;30:2636–2649. doi: 10.1523/JNEUROSCI.4456-09.2010. - DOI - PMC - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

