Pharmacokinetics and Safety of Vortioxetine in Pediatric Patients
- PMID: 28333546
- PMCID: PMC5568018
- DOI: 10.1089/cap.2016.0155
Pharmacokinetics and Safety of Vortioxetine in Pediatric Patients
Abstract
Objective: The primary objectives of this study were to evaluate the pharmacokinetics (PK) and tolerability of single and multiple doses of vortioxetine in children and adolescents with a depressive or anxiety disorder and to provide supportive information for appropriate dosing regimens for pediatric clinical trials.
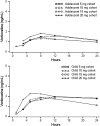
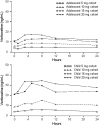
Methods: This prospective, open-label, multinational, multisite, multiple-dose trial enrolled 48 patients (children and adolescents; 1:1 ratio) divided into 8 cohorts (4 adolescent and 4 child), with each cohort including 6 patients. The cohorts in each age group were assigned to receive one of four dosing regimens: vortioxetine 5, 10, 15, or 20 mg q.d. for 14 days. The total treatment period lasted 14-20 days with patients in the higher dose cohorts uptitrated over 2-6 days. Plasma samples for PK analysis were obtained on the first and last days of dosing.
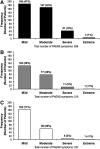
Results: Among children and adolescents, respectively, 62% and 92% had depression and 58% and 33% had anxiety disorder. Comorbid attention-deficit/hyperactivity disorder (ADHD) was present in 50% of children and 38% of adolescents. After 14 days q.d. at the target dose, the PK of vortioxetine concentrations was generally proportional to the dose in both age groups. Exposure, as assessed by maximum plasma concentrations and area under the plasma concentration-time curve from time 0 to 24 hours, was 30%-40% lower in adolescents than in children. There was no significant relationship between sex, height, or ADHD diagnosis and PK parameters. Most adverse events were mild in severity and consistent with those seen in adults.
Conclusion: The results suggest that the dosages of vortioxetine evaluated (5-20 mg q.d.; approved for treatment in adults) and the uptitration schedule used are appropriate for pediatric efficacy and safety trials.
Keywords: adolescents; adverse events; antidepressant; children; dosing; pharmacokinetics; vortioxetine.
Conflict of interest statement
Dr. Findling receives or has received research support, acted as a consultant, and/or served on a speaker's bureau for Alcobra, American Academy of Child & Adolescent Psychiatry, American Physician Institute, American Psychiatric Press, Bracket, CogCubed, Cognition Group, Coronado Biosciences, Dana Foundation, Elsevier, Forest, Guilford Press, Ironshore, Johns Hopkins University Press, Jubilant Clinsys, KemPharm, Lundbeck, Merck, NIH, Neurim, Novartis, Otsuka, Oxford University Press, Pfizer, Physicians Postgraduate Press, Purdue, Rhodes Pharmaceuticals, Roche, Sage, Shire, Sunovion, Supernus Pharmaceuticals, Transcept Pharmaceuticals, Tris, Validus, and WebMD. Dr. Robb receives or has received research support, acted as a consultant, received travel support, and/or served on a speaker's bureau for American Academy of Child & Adolescent Psychiatry, American Academy of Pediatrics, American Academy of Osteopathic Pediatricians, Allergan/Actavis/Forest, Bracket, CME Outfitters, Eli Lilly, GlaxoSmithKline, Guilford Press, Ironshore, Johnson & Johnson/Janssen, Lundbeck, Merck, NIH, Neuronetics, Otsuka, Pfizer, Rhodes Pharmaceuticals, Society of Maternal and Fetal Medicine, Sunovion, Supernus, SyneuRx, Takeda, and Tris. Dr. DelBello receives or has received research support, acted as a consultant, received travel support, and/or served on a speaker's bureau for American Academy of Child & Adolescent Psychiatry, Actavis, Bracket, Eli Lilly, GlaxoSmithKline, Guilford Press, Johnson & Johnson, Lundbeck, Pfizer, Purdue, Shire, Sunovion, Supernus, and Takeda. Dr. Michael Huss receives or has received research support, acted as a consultant, received travel support, and/or served on a speaker's bureau for Actelion, Eli Lilly, Englehardt, Janssen-Cilaq, Lundbeck, Medice, Novartis, Shire, Steiner-Arzneimittel, and has an international patent on Doppler radar to assess ADHD (Government Research Grants: BMBF, BMFT, BfArM). Dr. McNamara receives or has received research support from Forest, Lundbeck, Otsuka, National Institute of Mental Health, Roche, Sunovion, Shire, and Tonix, and has received honorarium for participation and travel support for a meeting from Sunovion. Dr. Sarkis receives or has received research support from Alcobra, Alkermes, Allergan, Assurex, Auspexm, Daiichi-Sankyo, Eli-Lilly and Company, Forest Pharmaceuticals, H. Lundbeck A/S, Indivior, Ironshore, H. Lundbeck, Medgenics, Otsuka, Palatin, Pfizer, Polus, Sage, Shire, Sunovion, Supernus, Takeda, Tal Medical, TEVA, and Tonix. Dr. Scheffer has no conflicts of interest and has received no stipend or any other payment. Dr. Poulsen is an employee of H. Lundbeck A/S. Dr. Chen is an employee of Takeda Pharmaceuticals Company, Ltd. Dr. Areberg is an employee of H. Lundbeck A/S. Mr. Ole Michael Lemming is an employee of H. Lundbeck A/S. At the time of this study, Dr. Auby was an employee of H. Lundbeck A/S.
Figures

References
-
- Bang-Andersen B, Ruhland T, Jorgensen M, Smith G, Frederiksen K, Jensen KG, Zhong H, Nielsen SM, Hogg S, Mork A, Stensbol TB: Discovery of 1-[2-(2,4-dimethylphenylsulfanyl)phenyl]piperazine (Lu AA21004): A novel multimodal compound for the treatment of major depressive disorder. J Med Chem 54:3206–3221, 2011 - PubMed
-
- Bidzan L, Mahableshwarkar AR, Jacobsen P, Yan M, Sheehan DV: Vortioxetine (Lu AA21004) in generalized anxiety disorder: Results of an 8-week, multinational, randomized, double-blind, placebo-controlled clinical trial. Eur Neuropsychopharmacol 22:847–857, 2012 - PubMed
-
- Birmaher B, Brent D, Bernet W, Bukstein O, Walter H, Benson RS, Chrisman A, Farchione T, Greenhill L, Hamilton J, Keable H, Kinlan J, Schoettle U, Stock S, Ptakowski KK, Medicus J: Practice parameter for the assessment and treatment of children and adolescents with depressive disorders. J Am Acad Child Adolesc Psychiatry 46:1503–1526, 2007 - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

