Cationic Nanoparticles Assembled from Natural-Based Steroid Lipid for Improved Intracellular Transport of siRNA and pDNA
- PMID: 28335197
- PMCID: PMC5302561
- DOI: 10.3390/nano6040069
Cationic Nanoparticles Assembled from Natural-Based Steroid Lipid for Improved Intracellular Transport of siRNA and pDNA
Abstract
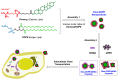
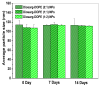
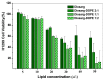
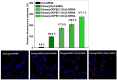
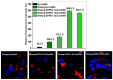
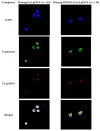
Developing new functional biomaterials from biocompatible natural-based resources for gene/drug delivery has attracted increasing attention in recent years. In this work, we prepared a series of cationic nanoparticles (Diosarg-DOPE NPs) by assembly of a natural steroid diosgenin-based cationic lipid (Diosarg) with commercially-available helper lipid 1,2-dioleoyl-sn-glycero-3-phosphorethanolamine (DOPE). These cationic Diosarg-DOPE NPs were able to efficiently bind siRNA and plasmid DNA (pDNA) via electrostatic interactions to form stable, nano-sized cationic lipid nanoparticles instead of lamellar vesicles in aqueous solution. The average particle size, zeta potentials and morphologies of the siRNA and pDNA complexes of the Diosarg-DOPE NPs were examined. The in vitro cytotoxicity of NPs depends on the dose and assembly ratio of the Diosarg and DOPE. Notably, the intracellular transportation efficacy of the exogenesis siRNA and pDNA could be greatly improved by using the Diosarg-DOPE NPs as the cargoes in H1299 cell line. The results demonstrated that the self-assembled Diosarg-DOPE NPs could achieve much higher intracellular transport efficiency for siRNA or pDNA than the cationic lipid Diosarg, indicating that the synergetic effect of different functional lipid components may benefit the development of high efficiency nano-scaled gene carriers. Moreover, it could be noted that the traditional "lysosome localization" involved in the intracellular trafficking of the Diosarg and Diosarg-DOPE NPs, indicating the co-assembly of helper lipid DOPE, might not significantly affect the intracellular localization features of the cationic lipids.
Keywords: diosgenin; intracellular uptake; pDNA; siRNA; steroid.
Conflict of interest statement
The authors declare no conflict of interest.
Figures



Similar articles
-
Skeleton-Controlled pDNA Delivery of Renewable Steroid-Based Cationic Lipids, the Endocytosis Pathway Analysis and Intracellular Localization.Int J Mol Sci. 2018 Jan 26;19(2):369. doi: 10.3390/ijms19020369. Int J Mol Sci. 2018. PMID: 29373505 Free PMC article.
-
How does the spacer length of cationic gemini lipids influence the lipoplex formation with plasmid DNA? Physicochemical and biochemical characterizations and their relevance in gene therapy.Biomacromolecules. 2012 Dec 10;13(12):3926-37. doi: 10.1021/bm301066w. Epub 2012 Nov 16. Biomacromolecules. 2012. PMID: 23130552
-
DC-Chol/DOPE cationic liposomes: a comparative study of the influence factors on plasmid pDNA and siRNA gene delivery.Int J Pharm. 2010 May 10;390(2):198-207. doi: 10.1016/j.ijpharm.2010.01.035. Epub 2010 Jan 29. Int J Pharm. 2010. PMID: 20116418
-
Ca(2+)-mediated anionic lipid-plasmid DNA lipoplexes. Electrochemical, structural, and biochemical studies.Langmuir. 2014 Oct 7;30(39):11704-13. doi: 10.1021/la502823z. Epub 2014 Sep 25. Langmuir. 2014. PMID: 25211646
-
Natural steroid-based cationic copolymers cholesterol/diosgenin-r-PDMAEMAs and their pDNA nanoplexes: impact of steroid structures and hydrophobic/hydrophilic ratios on pDNA delivery.RSC Adv. 2021 Jun 1;11(32):19450-19460. doi: 10.1039/d1ra00223f. eCollection 2021 May 27. RSC Adv. 2021. PMID: 35479247 Free PMC article.
Cited by
-
Research Progress on Liposome Pulmonary Delivery of Mycobacterium tuberculosis Nucleic Acid Vaccine and Its Mechanism of Action.J Aerosol Med Pulm Drug Deliv. 2024 Oct;37(5):284-298. doi: 10.1089/jamp.2023.0025. Epub 2024 Apr 26. J Aerosol Med Pulm Drug Deliv. 2024. PMID: 38669118 Review.
-
The effect of polar headgroups and spacer length on the DNA transfection of cholesterol-based cationic lipids.RSC Med Chem. 2020 Jan 6;11(2):212-224. doi: 10.1039/c9md00459a. eCollection 2020 Feb 1. RSC Med Chem. 2020. PMID: 33479628 Free PMC article.
-
Enzyme-Responsive Materials as Carriers for Improving Photodynamic Therapy.Front Chem. 2021 Nov 2;9:763057. doi: 10.3389/fchem.2021.763057. eCollection 2021. Front Chem. 2021. PMID: 34796163 Free PMC article. Review.
References
LinkOut - more resources
Full Text Sources
Other Literature Sources

