Cholesterol-Modified Amino-Pullulan Nanoparticles as a Drug Carrier: Comparative Study of Cholesterol-Modified Carboxyethyl Pullulan and Pullulan Nanoparticles
- PMID: 28335293
- PMCID: PMC5224631
- DOI: 10.3390/nano6090165
Cholesterol-Modified Amino-Pullulan Nanoparticles as a Drug Carrier: Comparative Study of Cholesterol-Modified Carboxyethyl Pullulan and Pullulan Nanoparticles
Abstract
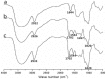
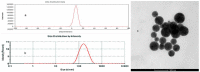
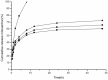
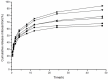
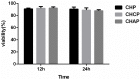
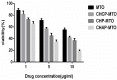
To search for nano-drug preparations with high efficiency in tumor treatment, we evaluated the drug-loading capacity and cell-uptake toxicity of three kinds of nanoparticles (NPs). Pullulan was grafted with ethylenediamine and hydrophobic groups to form hydrophobic cholesterol-modified amino-pullulan (CHAP) conjugates. Fourier transform infrared spectroscopy and nuclear magnetic resonance were used to identify the CHAP structure and calculate the degree of substitution of the cholesterol group. We compared three types of NPs with close cholesterol hydrophobic properties: CHAP, cholesterol-modified pullulan (CHP), and cholesterol-modified carboxylethylpullulan (CHCP), with the degree of substitution of cholesterol of 2.92%, 3.11%, and 3.46%, respectively. As compared with the two other NPs, CHAP NPs were larger, 263.9 nm, and had a positive surface charge of 7.22 mV by dynamic light-scattering measurement. CHAP NPs showed low drug-loading capacity, 12.3%, and encapsulation efficiency of 70.8%, which depended on NP hydrophobicity and was affected by surface charge. The drug release amounts of all NPs increased in the acid media, with CHAP NPs showing drug-release sensitivity with acid change. Cytotoxicity of HeLa cells was highest with mitoxantrone-loaded CHAP NPs on MTT assay. CHAP NPs may have potential as a high-efficiency drug carrier for tumor treatment.
Keywords: amino pullulan; cytotoxicity; degree of substitution; drug release; surface charge.
Conflict of interest statement
The authors declare no conflict of interest.
Figures


References
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

