Visualization of the Genomic Loci That Are Bound by Specific Multiprotein Complexes by Bimolecular Fluorescence Complementation Analysis on Drosophila Polytene Chromosomes
- PMID: 28336073
- PMCID: PMC5542922
- DOI: 10.1016/bs.mie.2017.02.003
Visualization of the Genomic Loci That Are Bound by Specific Multiprotein Complexes by Bimolecular Fluorescence Complementation Analysis on Drosophila Polytene Chromosomes
Abstract
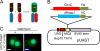
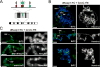
We have developed a procedure that enables visualization of the genomic loci that are bound by complexes formed by a specific combination of chromatin-binding proteins. This procedure is based on imaging bimolecular fluorescence complementation (BiFC) complexes on Drosophila polytene chromosomes. BiFC complexes are formed by the facilitated association of two fluorescent protein fragments that are fused to proteins that interact with, or are in close proximity to, each other. The intensity of BiFC complex fluorescence at individual genomic loci is greatly enhanced by the parallel alignment of hundreds of chromatids within the polytene chromosomes. The loci that are bound by the complexes are mapped by comparing the locations of BiFC complex fluorescence with the stereotypical banding patterns of the chromosomes. We describe strategies for the design, expression, and validation of fusion proteins for the analysis of BiFC complex binding on polytene chromosomes. We detail protocols for the preparation of polytene chromosome spreads that have been optimized for the purpose of BiFC complex visualization. Finally, we provide guidance for the interpretation of results from studies of BiFC complex binding on polytene chromosomes and for comparison of the genomic loci that are bound by BiFC complexes with those that are bound by subunits of the corresponding endogenous complexes. The visualization of BiFC complex binding on polytene chromosomes provides a unique method to visualize multiprotein complex binding at specific loci, throughout the genome, in individual cells.
Keywords: BiFC complex imaging; Chromatin binding proteins; Fluorescent protein fragments; Genomewide analysis; Polytene chromosome spreads; Transcription factor interaction; Transgene expression in specific tissues.
© 2017 Elsevier Inc. All rights reserved.
Figures

Similar articles
-
Determination of Complex Formation between Drosophila Nrf2 and GATA4 Factors at Selective Chromatin Loci Demonstrates Transcription Coactivation.Cells. 2023 Mar 19;12(6):938. doi: 10.3390/cells12060938. Cells. 2023. PMID: 36980279 Free PMC article.
-
Visualization of the Drosophila dKeap1-CncC interaction on chromatin illumines cooperative, xenobiotic-specific gene activation.Development. 2014 Aug;141(16):3277-88. doi: 10.1242/dev.110528. Epub 2014 Jul 25. Development. 2014. PMID: 25063457 Free PMC article.
-
Bimolecular fluorescence complementation analysis of inducible protein interactions: effects of factors affecting protein folding on fluorescent protein fragment association.J Mol Biol. 2009 Dec 4;394(3):391-409. doi: 10.1016/j.jmb.2009.08.069. Epub 2009 Sep 3. J Mol Biol. 2009. PMID: 19733184 Free PMC article.
-
Visualization of molecular interactions using bimolecular fluorescence complementation analysis: characteristics of protein fragment complementation.Chem Soc Rev. 2009 Oct;38(10):2876-86. doi: 10.1039/b909638h. Epub 2009 Sep 4. Chem Soc Rev. 2009. PMID: 19771334 Free PMC article. Review.
-
Banding Pattern of Polytene Chromosomes as a Representation of Universal Principles of Chromatin Organization into Topological Domains.Biochemistry (Mosc). 2018 Apr;83(4):338-349. doi: 10.1134/S0006297918040053. Biochemistry (Mosc). 2018. PMID: 29626921 Review.
Cited by
-
Merkel cell polyomavirus protein ALTO modulates TBK1 activity to support persistent infection.PLoS Pathog. 2024 Jul 29;20(7):e1012170. doi: 10.1371/journal.ppat.1012170. eCollection 2024 Jul. PLoS Pathog. 2024. PMID: 39074144 Free PMC article.
-
Biophysical Techniques for Target Validation and Drug Discovery in Transcription-Targeted Therapy.Int J Mol Sci. 2020 Mar 26;21(7):2301. doi: 10.3390/ijms21072301. Int J Mol Sci. 2020. PMID: 32225120 Free PMC article. Review.
-
Determination of Complex Formation between Drosophila Nrf2 and GATA4 Factors at Selective Chromatin Loci Demonstrates Transcription Coactivation.Cells. 2023 Mar 19;12(6):938. doi: 10.3390/cells12060938. Cells. 2023. PMID: 36980279 Free PMC article.
References
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

