Heteroleptic Copper(I) Complexes of "Scorpionate" Bis-pyrazolyl Carboxylate Ligand with Auxiliary Phosphine as Potential Anticancer Agents: An Insight into Cytotoxic Mode
- PMID: 28338061
- PMCID: PMC5364558
- DOI: 10.1038/srep45229
Heteroleptic Copper(I) Complexes of "Scorpionate" Bis-pyrazolyl Carboxylate Ligand with Auxiliary Phosphine as Potential Anticancer Agents: An Insight into Cytotoxic Mode
Abstract
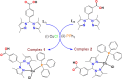
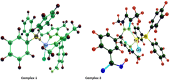
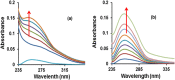
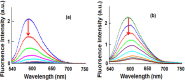
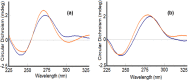
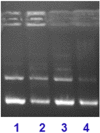
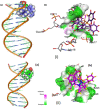
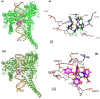
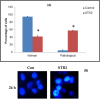
New copper(I) complexes [CuCl(PPh3)(L)] (1: L = LA = 4-carboxyphenyl)bis(3,5-dimethylpyrazolyl)methane; (2: L = LB = 3-carboxyphenyl)bis(3,5-dimethylpyrazolyl)methane) were prepared and characterised by elemental analysis and various spectroscopic techniques such as FT-IR, NMR, UV-Vis, and ESI-MS. The molecular structures of complexes 1 and 2 were analyzed by theoretical B3LYP/DFT method. Furthermore, in vitro DNA binding studies were carried out to check the ability of complexes 1 and 2 to interact with native calf thymus DNA (CT-DNA) using absorption titration, fluorescence quenching and circular dichroism, which is indicative of more avid binding of the complex 1. Moreover, DNA mobility assay was also conducted to study the concentration-dependent cleavage pattern of pBR322 DNA by complex 1, and the role of ROS species to have a mechanistic insight on the cleavage pattern, which ascertained substantial roles by both hydrolytic and oxidative pathways. Additionally, we analyzed the potential of the interaction of complex 1 with DNA and enzyme (Topo I and II) with the aid of molecular modeling. Furthermore, cytotoxic activity of complex 1 was tested against HepG2 cancer cell lines. Thus, the potential of the complex 1 is promising though further in vivo investigations may be required before subjecting it to clinical trials.
Conflict of interest statement
The authors declare no competing financial interests.
Figures



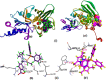
References
-
- World Cancer Report 2014, World Health Organization, pp. Chapter 1.1. and 5.6. ISBN 9283204298 (2014).
-
- Siegel R. L., Miller K. D. & Jemal A. Cancer Statistics, 2016. CA Cancer J. Clinicians, 66, 7–30 (2016). - PubMed
-
- Rosenberg B., VanCamp L., Trosko J. E. & Mansour V. H. Platinum compounds: a new class of potent antitumour agents. Nature, 222, 385–386 (1969). - PubMed
-
- Rosenberg B. Platinum complexes for the treatment of cancer. Interdiscip. Sci. Rev. 3, 134–147 (1978).
-
- Galanski M., Arion V. B., Jakupec M. A. & Keppler B. K. Recent developments in the field of tumor-inhibiting metal complexes Curr. Pharm. Des. 9, 2078–2089 (2003). - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

