A phase I trial of the MEK inhibitor selumetinib (AZD6244) in pediatric patients with recurrent or refractory low-grade glioma: a Pediatric Brain Tumor Consortium (PBTC) study
- PMID: 28339824
- PMCID: PMC5570236
- DOI: 10.1093/neuonc/now282
A phase I trial of the MEK inhibitor selumetinib (AZD6244) in pediatric patients with recurrent or refractory low-grade glioma: a Pediatric Brain Tumor Consortium (PBTC) study
Abstract
Background: Activation of the mitogen-activated protein kinase pathway is important for growth of pediatric low-grade gliomas (LGGs). The aim of this study was to determine the recommended phase II dose (RP2D) and the dose-limiting toxicities (DLTs) of the MEK inhibitor selumetinib in children with progressive LGG.
Methods: Selumetinib was administered orally starting at 33 mg/m2/dose b.i.d., using the modified continual reassessment method. Pharmacokinetic analysis was performed during the first course. BRAF aberrations in tumor tissue were determined by real-time polymerase chain reaction and fluorescence in situ hybridization.
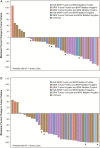
Results: Thirty-eight eligible subjects were enrolled. Dose levels 1 and 2 (33 and 43 mg/m2/dose b.i.d.) were excessively toxic. DLTs included grade 3 elevated amylase/lipase (n = 1), headache (n = 1), mucositis (n = 2), and grades 2-3 rash (n = 6). At dose level 0 (25 mg/m2/dose b.i.d, the RP2D), only 3 of 24 subjects experienced DLTs (elevated amylase/lipase, rash, and mucositis). At the R2PD, the median (range) area under the curve (AUC0-∞) and apparent oral clearance of selumetinib were 3855 ng*h/mL (1780 to 7250 ng × h/mL) and 6.5 L × h-1 × m-2 (3.4 to 14.0 L × h-1 × m-2), respectively. Thirteen of 19 tumors had BRAF abnormalities. Among the 5 (20%) of 25 subjects with sustained partial responses, all at the RP2D, 4 had BRAF aberrations, 1 had insufficient tissue. Subjects received a median of 13 cycles (range: 1-26). Fourteen (37%) completed all protocol treatment (26 cycles [n = 13], 13 cycles [n = 1]) with at least stable disease; 2-year progression-free survival at the RP2D was 69 ± SE 9.8%.
Conclusion: Selumetinib has promising antitumor activity in children with LGG. Rash and mucositis were the most common DLTs.
Keywords: low-grade glioma; phase I trial; selumetinib.
© The Author(s) 2017. Published by Oxford University Press on behalf of the Society for Neuro-Oncology. All rights reserved. For permissions, please e-mail: journals.permissions@oup.com.
Figures
References
-
- Tacke U, Karger D, Spreer J, et al. Incidence of vasculopathy in children with hypothalamic/chiasmatic gliomas treated with brachytherapy. Childs Nerv Syst. 2011;27(6):961–966. - PubMed
-
- Perkins SM, Fei W, Mitra N, et al. Late causes of death in children treated for CNS malignancies. J Neurooncol. 2013;115(1):79–85. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials