Glutathione species and metabolomic prints in subjects with liver disease as biological markers for the detection of hepatocellular carcinoma
- PMID: 28340971
- PMCID: PMC5144552
- DOI: 10.1016/j.hpb.2016.09.007
Glutathione species and metabolomic prints in subjects with liver disease as biological markers for the detection of hepatocellular carcinoma
Abstract
Background: The incidence of liver disease is increasing in USA. Animal models had shown glutathione species in plasma reflects liver glutathione state and it could be a surrogate for the detection of hepatocellular carcinoma (HCC).
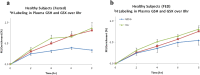
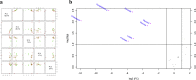
Methods: The present study aimed to translate methods to the human and to explore the role of glutathione/metabolic prints in the progression of liver dysfunction and in the detection of HCC. Treated plasma from healthy subjects (n = 20), patients with liver disease (ESLD, n = 99) and patients after transplantation (LTx, n = 7) were analyzed by GC- or LC/MS. Glutathione labeling profile was measured by isotopomer analyzes of 2H2O enriched plasma. Principal Component Analyzes (PCA) were used to determined metabolic prints.
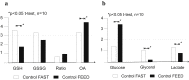
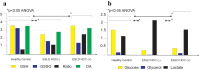
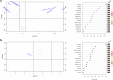
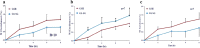
Results: There was a significant difference in glutathione/metabolic profiles from patients with ESLD vs healthy subjects and patients after LTx. Similar significant differences were noted on patients with ESLD when stratified by the MELD score. PCA analyses showed myristic acid, citric acid, succinic acid, l-methionine, d-threitol, fumaric acid, pipecolic acid, isoleucine, hydroxy-butyrate and glycolic, steraric and hexanoic acids were discriminative metabolites for ESLD-HCC+ vs ESLD-HCC- subject status.
Conclusions: Glutathione species and metabolic prints defined liver disease severity and may serve as surrogate for the detection of HCC in patients with established cirrhosis.
Copyright © 2016 The Authors. Published by Elsevier Ltd.. All rights reserved.
Figures

Similar articles
-
Relationships between blood levels of fat soluble vitamins and disease etiology and severity in adults awaiting liver transplantation.J Gastroenterol Hepatol. 2011 Sep;26(9):1402-10. doi: 10.1111/j.1440-1746.2011.06746.x. J Gastroenterol Hepatol. 2011. PMID: 21501225
-
NMR and LC/MS-based global metabolomics to identify serum biomarkers differentiating hepatocellular carcinoma from liver cirrhosis.Int J Cancer. 2014 Aug 1;135(3):658-68. doi: 10.1002/ijc.28706. Epub 2014 Jan 17. Int J Cancer. 2014. PMID: 24382646
-
Integrated metabolomic profiling of hepatocellular carcinoma in hepatitis C cirrhosis through GC/MS and UPLC/MS-MS.Liver Int. 2014 Oct;34(9):1428-44. doi: 10.1111/liv.12541. Epub 2014 Apr 28. Liver Int. 2014. PMID: 24661807 Free PMC article.
-
Liver transplantation for hepatocellular carcinoma.Curr Gastroenterol Rep. 2015 Mar;17(3):11. doi: 10.1007/s11894-015-0435-3. Curr Gastroenterol Rep. 2015. PMID: 25740250 Review.
-
Hepatocellular Carcinoma in Paediatric Patients with Alagille Syndrome: Case Series and Review of Literature.J Gastrointest Cancer. 2020 Sep;51(3):1047-1052. doi: 10.1007/s12029-020-00391-2. J Gastrointest Cancer. 2020. PMID: 32180165 Review. No abstract available.
Cited by
-
Targeting LRRC41 as a potential therapeutic approach for hepatocellular carcinoma.Front Mol Biosci. 2023 Dec 21;10:1300294. doi: 10.3389/fmolb.2023.1300294. eCollection 2023. Front Mol Biosci. 2023. PMID: 38192337 Free PMC article.
-
The Prognostic Role of Glutathione and Its Related Antioxidant Enzymes in the Recurrence of Hepatocellular Carcinoma.Nutrients. 2021 Nov 14;13(11):4071. doi: 10.3390/nu13114071. Nutrients. 2021. PMID: 34836325 Free PMC article.
-
Characterization of Salivary and Plasma Metabolites as Biomarkers for HCC: A Pilot Study.Cancers (Basel). 2023 Sep 12;15(18):4527. doi: 10.3390/cancers15184527. Cancers (Basel). 2023. PMID: 37760495 Free PMC article.
-
The Current Status of the Liver Liquid Biopsy in MASH Related HCC: Overview and Future Directions.Biomolecules. 2023 Sep 9;13(9):1369. doi: 10.3390/biom13091369. Biomolecules. 2023. PMID: 37759769 Free PMC article. Review.
-
Deciphering hepatocellular carcinoma through metabolomics: from biomarker discovery to therapy evaluation.Cancer Manag Res. 2018 Apr 11;10:715-734. doi: 10.2147/CMAR.S156837. eCollection 2018. Cancer Manag Res. 2018. PMID: 29692630 Free PMC article. Review.
References
-
- Bunchorntavakul C., Reddy K.R. Diagnosis and management of overlap syndromes. Clin Liver Dis. 2015;19:81–97. - PubMed
-
- Roberts H.W., Utuama O.A., Klevens M., Teshale E., Hughes E., Jiles R. The contribution of viral hepatitis to the burden of chronic liver disease in the United States. Am J Gastroenterol. 2014;109:387–393. quiz 6, 94. - PubMed
-
- Fox R.K. When to consider liver transplant during the management of chronic liver disease. Med Clin North Am. 2014;98:153–168. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

