Repair of oxidatively induced DNA damage by DNA glycosylases: Mechanisms of action, substrate specificities and excision kinetics
- PMID: 28342455
- PMCID: PMC7451025
- DOI: 10.1016/j.mrrev.2017.02.001
Repair of oxidatively induced DNA damage by DNA glycosylases: Mechanisms of action, substrate specificities and excision kinetics
Abstract
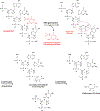
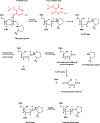
Endogenous and exogenous reactive species cause oxidatively induced DNA damage in living organisms by a variety of mechanisms. As a result, a plethora of mutagenic and/or cytotoxic products are formed in cellular DNA. This type of DNA damage is repaired by base excision repair, although nucleotide excision repair also plays a limited role. DNA glycosylases remove modified DNA bases from DNA by hydrolyzing the glycosidic bond leaving behind an apurinic/apyrimidinic (AP) site. Some of them also possess an accompanying AP-lyase activity that cleaves the sugar-phosphate chain of DNA. Since the first discovery of a DNA glycosylase, many studies have elucidated the mechanisms of action, substrate specificities and excision kinetics of these enzymes present in all living organisms. For this purpose, most studies used single- or double-stranded oligodeoxynucleotides with a single DNA lesion embedded at a defined position. High-molecular weight DNA with multiple base lesions has been used in other studies with the advantage of the simultaneous investigation of many DNA base lesions as substrates. Differences between the substrate specificities and excision kinetics of DNA glycosylases have been found when these two different substrates were used. Some DNA glycosylases possess varying substrate specificities for either purine-derived lesions or pyrimidine-derived lesions, whereas others exhibit cross-activity for both types of lesions. Laboratory animals with knockouts of the genes of DNA glycosylases have also been used to provide unequivocal evidence for the substrates, which had previously been found in in vitro studies, to be the actual substrates in vivo as well. On the basis of the knowledge gained from the past studies, efforts are being made to discover small molecule inhibitors of DNA glycosylases that may be used as potential drugs in cancer therapy.
Keywords: DNA glycosylases; DNA repair; Excision kinetics; Oxidatively induced DNA damage; Substrate specificities.
Published by Elsevier B.V.
Conflict of interest statement
Conflict of interest statement
The authors declare that there are no conflicts of interest.
Figures





References
-
- Halliwell B, Gutteridge JMC, Free Radicals in Biology and Medicine, Fifth Edition, Oxford University Press, Oxford, 2015.
-
- Beckman RA, Loeb LA, Genetic instability in cancer: theory and experiment, Semin. Cancer Biol 15 (2005) 423–435. - PubMed
-
- Hanahan D, Weinberg RA, The hallmarks of cancer, Cell 100 (2000) 57–70. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous

