Copper Nanowires and Their Applications for Flexible, Transparent Conducting Films: A Review
- PMID: 28344304
- PMCID: PMC5302514
- DOI: 10.3390/nano6030047
Copper Nanowires and Their Applications for Flexible, Transparent Conducting Films: A Review
Abstract
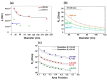
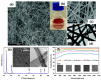
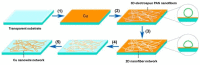
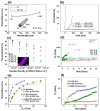
Cu nanowires (NWs) are attracting considerable attention as alternatives to Ag NWs for next-generation transparent conductors, replacing indium tin oxide (ITO) and micro metal grids. Cu NWs hold great promise for low-cost fabrication via a solution-processed route and show preponderant optical, electrical, and mechanical properties. In this study, we report a summary of recent advances in research on Cu NWs, covering the optoelectronic properties, synthesis routes, deposition methods to fabricate flexible transparent conducting films, and their potential applications. This review also examines the approaches on protecting Cu NWs from oxidation in air environments.
Keywords: ITO replacement; copper nanowires; flexible; optoelectronic properties; protection; transparent conducting films.
Conflict of interest statement
The authors declare no conflict of interest.
Figures



Similar articles
-
Solution-Processed Copper/Reduced-Graphene-Oxide Core/Shell Nanowire Transparent Conductors.ACS Nano. 2016 Feb 23;10(2):2600-6. doi: 10.1021/acsnano.5b07651. Epub 2016 Jan 28. ACS Nano. 2016. PMID: 26820809
-
Facile Synthesis of Silver Nanowires with Different Aspect Ratios and Used as High-Performance Flexible Transparent Electrodes.Nanoscale Res Lett. 2017 Dec;12(1):480. doi: 10.1186/s11671-017-2259-6. Epub 2017 Aug 7. Nanoscale Res Lett. 2017. PMID: 28789485 Free PMC article.
-
Synthesis of Copper Nanowires Using Monoethanolamine and the Application in Transparent Conductive Films.Nanomaterials (Basel). 2025 Apr 22;15(9):638. doi: 10.3390/nano15090638. Nanomaterials (Basel). 2025. PMID: 40358255 Free PMC article.
-
Recent progress of solution-processed Cu nanowires transparent electrodes and their applications.RSC Adv. 2019 Aug 28;9(46):26961-26980. doi: 10.1039/c9ra04404c. eCollection 2019 Aug 23. RSC Adv. 2019. PMID: 35528598 Free PMC article. Review.
-
Nanowire-based transparent conductors for flexible electronics and optoelectronics.Sci Bull (Beijing). 2017 Jan 30;62(2):143-156. doi: 10.1016/j.scib.2016.11.009. Epub 2016 Dec 7. Sci Bull (Beijing). 2017. PMID: 36659486 Review.
Cited by
-
Comprehensive Study on Chemical and Hot Press-Treated Silver Nanowires for Efficient Polymer Solar Cell Application.Polymers (Basel). 2017 Nov 22;9(11):635. doi: 10.3390/polym9110635. Polymers (Basel). 2017. PMID: 30965936 Free PMC article.
-
Anisotropic Growth of Copper Nanorods Mediated by Cl- Ions.ACS Omega. 2022 Feb 16;7(8):7414-7420. doi: 10.1021/acsomega.2c00359. eCollection 2022 Mar 1. ACS Omega. 2022. PMID: 35252731 Free PMC article.
-
Fabrication of air-stable, transparent Cu grid electrodes by etching through a PVA-based protecting layer patterned using a screen mesh.RSC Adv. 2018 Apr 19;8(27):14864-14869. doi: 10.1039/c7ra11966f. eCollection 2018 Apr 18. RSC Adv. 2018. PMID: 35541308 Free PMC article.
-
Emergence of Flexible White Organic Light-Emitting Diodes.Polymers (Basel). 2019 Feb 22;11(2):384. doi: 10.3390/polym11020384. Polymers (Basel). 2019. PMID: 30960368 Free PMC article. Review.
-
Highly Stable, Flexible, Transparent Hybrid Strontium Titanate Conductive Thin Films with Embedded Cu Nanowires.Materials (Basel). 2025 May 21;18(10):2398. doi: 10.3390/ma18102398. Materials (Basel). 2025. PMID: 40429135 Free PMC article.
References
-
- Kang M.G., Kim M.S., Kim J., Guo L.J. Organic solar cells using nanoimprinted transparent metal electrodes. Adv. Mater. 2008;20:4408–4413. doi: 10.1002/adma.200800750. - DOI
-
- Tak Y.-H., Kim K.-B., Park H.-G., Lee K.-H., Lee J.-R. Criteria for ITO (indium–tin-oxide) thin film as the bottom electrode of an organic light emitting diode. Thin Solid Films. 2002;411:12–16. doi: 10.1016/S0040-6090(02)00165-7. - DOI
-
- Loureiro J., Neves N., Barros R., Mateus T., Santos R., Filonovich S., Reparaz S., Sotomayor-Torres C.M., Wyczisk F., Divay L., et al. Transparent aluminium zinc oxide thin films with enhanced thermoelectric properties. J. Mater. Chem. A. 2014;2:6649–6655. doi: 10.1039/c3ta15052f. - DOI
Publication types
LinkOut - more resources
Full Text Sources
Other Literature Sources

