Metal-Catalysed Azidation of Organic Molecules
- PMID: 28344503
- PMCID: PMC5347896
- DOI: 10.1002/ejoc.201601390
Metal-Catalysed Azidation of Organic Molecules
Abstract
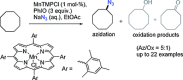
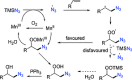
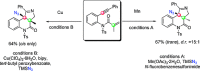
The azide moiety is a desirable functionality in organic molecules, useful in a variety of transformations such as olefin aziridination, C-H bond amination, isocyanate synthesis, the Staudinger reaction and the formation of azo compounds. To harness the versatility of the azide functionality fully it is important that these compounds be easy to prepare, in a clean and cost-effective manner. Conventional (non-catalysed) methods to synthesise azides generally require quite harsh reaction conditions that are often not tolerant of functional groups. In the last decade, several metal-catalysed azidations have been developed in attempts to circumvent this problem. These methods are generally faster, cleaner and more functional-group-tolerant than conventional methods to prepare azides, and can sometimes even be conveniently combined with one-pot follow-up transformations of the installed azide moiety. This review highlights metal-catalysed approaches to azide synthesis, with a focus on the substrate scopes and mechanisms, as well as on advantages and disadvantages of the methods. Overall, metal-catalysed azidation reactions provide shorter routes to a variety of potentially useful organic molecules containing the azide moiety.
Keywords: Azides; C–H activation; Noble‐metal catalysis; Radical reactions; Synthetic methods; Transition‐metal catalysis.
Figures












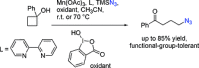







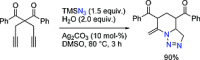
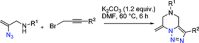
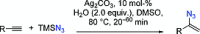
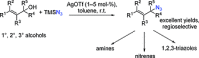
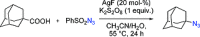
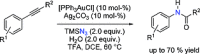



Similar articles
-
Metal-catalysed azidation of tertiary C-H bonds suitable for late-stage functionalization.Nature. 2015 Jan 29;517(7536):600-4. doi: 10.1038/nature14127. Nature. 2015. PMID: 25631448 Free PMC article.
-
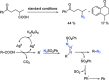
Transition-metal-catalyzed C-N bond forming reactions using organic azides as the nitrogen source: a journey for the mild and versatile C-H amination.Acc Chem Res. 2015 Apr 21;48(4):1040-52. doi: 10.1021/acs.accounts.5b00020. Epub 2015 Mar 30. Acc Chem Res. 2015. PMID: 25821998
-
From mechanism to mouse: a tale of two bioorthogonal reactions.Acc Chem Res. 2011 Sep 20;44(9):666-76. doi: 10.1021/ar200148z. Epub 2011 Aug 15. Acc Chem Res. 2011. PMID: 21838330 Free PMC article.
-
Electrophilic Azides for Materials Synthesis and Chemical Biology.Acc Chem Res. 2020 Apr 21;53(4):937-948. doi: 10.1021/acs.accounts.0c00046. Epub 2020 Mar 24. Acc Chem Res. 2020. PMID: 32207916 Review.
-
Azides in the Synthesis of Various Heterocycles.Molecules. 2022 Jun 9;27(12):3716. doi: 10.3390/molecules27123716. Molecules. 2022. PMID: 35744839 Free PMC article. Review.
Cited by
-
Decatungstate-photocatalysed C(sp3)-H azidation.Chem Commun (Camb). 2022 Apr 14;58(31):4869-4872. doi: 10.1039/d2cc00425a. Chem Commun (Camb). 2022. PMID: 35348566 Free PMC article.
-
Azido-alkynylation of alkenes through radical-polar crossover.Chem Sci. 2023 Aug 11;14(35):9452-9460. doi: 10.1039/d3sc03309k. eCollection 2023 Sep 13. Chem Sci. 2023. PMID: 37712015 Free PMC article.
-
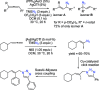
Mechanistic Investigation of the Iron-Catalyzed Azidation of Alkyl C(sp3)-H Bonds with Zhdankin's λ3-Azidoiodane.J Am Chem Soc. 2021 Oct 6;143(39):16184-16196. doi: 10.1021/jacs.1c07330. Epub 2021 Sep 24. J Am Chem Soc. 2021. PMID: 34559970 Free PMC article.
-
Selective Azidooxygenation of Alkenes Enabled by Photo-induced Radical Transfer Using Aryl-λ3-azidoiodane Species.ACS Omega. 2021 Sep 27;6(40):26623-26639. doi: 10.1021/acsomega.1c03991. eCollection 2021 Oct 12. ACS Omega. 2021. PMID: 34661016 Free PMC article.
-
Towards an Asymmetric Organocatalytic α-Azidation of β-Ketoesters.Molecules. 2018 May 11;23(5):1142. doi: 10.3390/molecules23051142. Molecules. 2018. PMID: 29751597 Free PMC article.
References
-
- Intrieri D., Zardi P., Caselli A. and Gallo E., Chem. Commun., 2014, 50, 11440–11453. - PubMed
-
- For a recent overview of metal‐catalysed click reactions, see: Wang C., Ikhlef D., Kahlal S., Saillard J.‐Y. and Astruc D., Coord. Chem. Rev., 2016, 316, 1–20.
-
- Bräse S., Gil C., Knepper K. and Zimmermann V., Angew. Chem. Int. Ed., 2005, 44, 5188–5240; - PubMed
- Angew. Chem., 2005, 117, 5320.
-
- Degennaro L., Trinchera P. and Luisi R., Chem. Rev., 2014, 114, 7881–7929. - PubMed
-
- a) van Kalkeren H. A., Bruins J. J., Rutjes F. P. J. T. and van Delft F. L., Adv. Synth. Catal., 2012, 354, 1417–1421;
- b) Takaoka A., Moret M. and Peters J. C., J. Am. Chem. Soc., 2012, 134, 6695–6706; - PubMed
- c) Bellow J. A., Yousif M., Cabelof A. C., Lord R. L. and Groysman S., Organometallics, 2015, 34, 2917–2923.
Publication types
LinkOut - more resources
Full Text Sources
Other Literature Sources
