Oncolytic Adenoviruses Armed with Tumor Necrosis Factor Alpha and Interleukin-2 Enable Successful Adoptive Cell Therapy
- PMID: 28345026
- PMCID: PMC5363700
- DOI: 10.1016/j.omto.2016.12.004
Oncolytic Adenoviruses Armed with Tumor Necrosis Factor Alpha and Interleukin-2 Enable Successful Adoptive Cell Therapy
Abstract
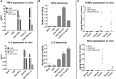
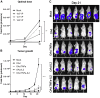
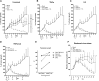
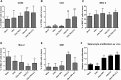
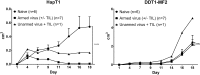
Adoptive cell therapy holds much promise in the treatment of cancer but results in solid tumors have been modest. The notable exception is tumor-infiltrating lymphocyte (TIL) therapy of melanoma, but this approach only works with high-dose preconditioning chemotherapy and systemic interleukin (IL)-2 postconditioning, both of which are associated with toxicities. To improve and broaden the applicability of adoptive cell transfer, we constructed oncolytic adenoviruses coding for human IL-2 (hIL2), tumor necrosis factor alpha (TNF-α), or both. The viruses showed potent antitumor efficacy against human tumors in immunocompromised severe combined immunodeficiency (SCID) mice. In immunocompetent Syrian hamsters, we combined the viruses with TIL transfer and were able to cure 100% of the animals. Cured animals were protected against tumor re-challenge, indicating a memory response. Arming with IL-2 and TNF-α increased the frequency of both CD4+ and CD8+ TILs in vivo and augmented splenocyte proliferation ex vivo, suggesting that the cytokines were important for T cell persistence and proliferation. Cytokine expression was limited to tumors and treatment-related signs of systemic toxicity were absent, suggesting safety. To conclude, cytokine-armed oncolytic adenoviruses enhanced adoptive cell therapy by favorable alteration of the tumor microenvironment. A clinical trial is in progress to study the utility of Ad5/3-E2F-d24-hTNFa-IRES-hIL2 (TILT-123) in human patients with cancer.
Keywords: TNF; adenovirus; cytokines; interleukin 2; tumor-infiltrating lymphocytes.
Figures

References
-
- Rosenberg S.A., Yang J.C., Sherry R.M., Kammula U.S., Hughes M.S., Phan G.Q., Citrin D.E., Restifo N.P., Robbins P.F., Wunderlich J.R. Durable complete responses in heavily pretreated patients with metastatic melanoma using T-cell transfer immunotherapy. Clin. Cancer Res. 2011;17:4550–4557. - PMC - PubMed
-
- Andtbacka R.H., Kaufman H.L., Collichio F., Amatruda T., Senzer N., Chesney J., Delman K.A., Spitler L.E., Puzanov I., Agarwala S.S. Talimogene laherparepvec improves durable response rate in patients with advanced melanoma. J. Clin. Oncol. 2015;33:2780–2788. - PubMed
-
- Li H., Wang C., Yu J., Cao S., Wei F., Zhang W., Han Y., Ren X.B. Dendritic cell-activated cytokine-induced killer cells enhance the anti-tumor effect of chemotherapy on non-small cell lung cancer in patients after surgery. Cytotherapy. 2009;11:1076–1083. - PubMed
-
- Zhao X., Zhang Z., Li H., Huang J., Yang S., Xie T., Huang L., Yue D., Xu L., Wang L. Cytokine induced killer cell-based immunotherapies in patients with different stages of renal cell carcinoma. Cancer Lett. 2015;362:192–198. - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

