7-year follow-up after stereotactic ablative radiotherapy for patients with stage I non-small cell lung cancer: Results of a phase 2 clinical trial
- PMID: 28346656
- PMCID: PMC5544582
- DOI: 10.1002/cncr.30693
7-year follow-up after stereotactic ablative radiotherapy for patients with stage I non-small cell lung cancer: Results of a phase 2 clinical trial
Abstract
Background: The authors evaluated the efficacy, patterns of failure, and toxicity of stereotactic ablative radiotherapy (SABR) for patients with medically inoperable, clinical stage I non-small cell lung cancer (NSCLC) in a prospective clinical trial with 7 years of follow-up. Clinical staging was performed according to the seventh edition of the American Joint Committee on Cancer TNM staging system.
Methods: Eligible patients with histologically confirmed NSCLC of clinical stage I as determined using positron emission tomography staging were treated with SABR (50 grays in 4 fractions). The primary endpoint was progression-free survival. Patients were followed with computed tomography and/or positron emission tomography/computed tomography every 3 months for the first 2 years, every 6 months for the next 3 years, and then annually thereafter.
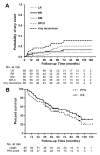
Results: A total of 65 patients were eligible for analysis. The median age of the patients was 71 years, and the median follow-up was 7.2 years. A total of 18 patients (27.7%) developed disease recurrence at a median of 14.5 months (range, 4.3-71.5 months) after SABR. Estimated incidences of local, regional, and distant disease recurrence using competing risk analysis were 8.1%, 10.9%, and 11.0%, respectively, at 5 years and 8.1%, 13.6%, and 13.8%, respectively, at 7 years. A second primary lung carcinoma developed in 12 patients (18.5%) at a median of 35 months (range, 5-67 months) after SABR. Estimated 5-year and 7-year progression-free survival rates were 49.5% and 38.2%, respectively; the corresponding overall survival rates were 55.7% and 47.5%, respectively. Three patients (4.6%) experienced grade 3 treatment-related adverse events. No patients developed grade 4 or 5 adverse events (toxicity was graded according to the National Cancer Institute Common Terminology Criteria for Adverse Events [version 3.0]).
Conclusions: With long-term follow-up, the results of the current prospective study demonstrated outstanding local control and low toxicity after SABR in patients with clinical stage I NSCLC. Regional disease recurrence and distant metastases were the dominant manifestations of failure. Surveillance for second primary lung carcinoma is recommended. Cancer 2017;123:3031-39. © 2017 American Cancer Society.
Keywords: non-small cell lung cancer (NSCLC); patterns of failure; stage I disease; stereotactic ablative radiotherapy (SABR); stereotactic body radiotherapy.
© 2017 American Cancer Society.
Conflict of interest statement
Figures

Comment in
-
Encouraging long-term outcomes reported in patients with stage I non-small cell lung cancer treated with stereotactic ablative radiotherapy.CA Cancer J Clin. 2017 Sep;67(5):349-350. doi: 10.3322/caac.21375. Epub 2017 Jul 11. CA Cancer J Clin. 2017. PMID: 28697271 No abstract available.
References
-
- Crabtree TD, Denlinger CE, Meyers BF, et al. Stereotactic body radiation therapy versus surgical resection for stage I non-small cell lung cancer. J Thorac Cardiovasc Surg. 2010;140:377–386. - PubMed
-
- Verstegen NE, Oosterhuis JW, Palma DA, et al. Stage I-II non-small-cell lung cancer treated using either stereotactic ablative radiotherapy (SABR) or lobectomy by video-assisted thoracoscopic surgery (VATS): outcomes of a propensity score-matched analysis. Ann Oncol. 2013;24:1543–1548. - PubMed
-
- Port JL, Parashar B, Osakwe N, et al. A propensity-matched analysis of wedge resection and stereotactic body radiotherapy for early stage lung cancer. Ann Thorac Surg. 2014;98:1152–1159. - PubMed
-
- Chang JY, Bezjak A, Mornex F. Stereotactic ablative radiotherapy for centrally located early stage non-small-cell lung cancer: what we have learned. J Thorac Oncol. 2015;10:577–585. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

