Craniofacial Tissue Engineering
- PMID: 28348178
- PMCID: PMC5749148
- DOI: 10.1101/cshperspect.a025775
Craniofacial Tissue Engineering
Abstract
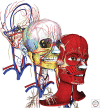
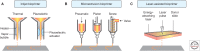
The craniofacial complex is composed of fundamental components such as blood vessels and nerves, and also a variety of specialized tissues such as craniofacial bones, cartilages, muscles, ligaments, and the highly specialized and unique organs, the teeth. Together, these structures provide many functions including speech, mastication, and aesthetics of the craniofacial complex. Craniofacial defects not only influence the structure and function of the jaws and face, but may also result in deleterious psychosocial issues, emphasizing the need for rapid and effective, precise, and aesthetic reconstruction of craniofacial tissues. In a broad sense, craniofacial tissue reconstructions share many of the same issues as noncraniofacial tissue reconstructions. Therefore, many concepts and therapies for general tissue engineering can and have been used for craniofacial tissue regeneration. Still, repair of craniofacial defects presents unique challenges, mainly because of their complex and unique 3D geometry.
Copyright © 2018 Cold Spring Harbor Laboratory Press; all rights reserved.
Figures

References
-
- Abukawa H, Zhang W, Young CS, Asrican R, Vacanti JP, Kaban LB, Troulis MJ, Yelick PC. 2009. Reconstructing mandibular defects using autologous tissue-engineered tooth and bone constructs. J Oral Maxillofac Surg 67: 335–347. - PubMed
-
- Abzhanov A, Rodda SJ, McMahon AP, Tabin CJ. 2007. Regulation of skeletogenic differentiation in cranial dermal bone. Development 134: 3133–3144. - PubMed
-
- Agata H, Watanabe N, Ishii Y, Kubo N, Ohshima S, Yamazaki M, Tojo A, Kagami H. 2009. Feasibility and efficacy of bone tissue engineering using human bone marrow stromal cells cultivated in serum-free conditions. Biochem Biophys Res Commun 382: 353–358. - PubMed
-
- Akintoye SO, Lam T, Shi S, Brahim J, Collins MT, Robey PG. 2006. Skeletal site-specific characterization of orofacial and iliac crest human bone marrow stromal cells in same individuals. Bone 38: 758–768. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
