NLRP3 Deficiency Attenuates Renal Fibrosis and Ameliorates Mitochondrial Dysfunction in a Mouse Unilateral Ureteral Obstruction Model of Chronic Kidney Disease
- PMID: 28348462
- PMCID: PMC5350413
- DOI: 10.1155/2017/8316560
NLRP3 Deficiency Attenuates Renal Fibrosis and Ameliorates Mitochondrial Dysfunction in a Mouse Unilateral Ureteral Obstruction Model of Chronic Kidney Disease
Abstract
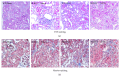
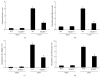
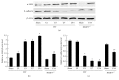
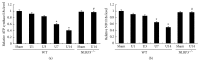
Background and Aims. The nucleotide-binding domain and leucine-rich repeat containing PYD-3 (NLRP3) inflammasome has been implicated in the pathogenesis of chronic kidney disease (CKD); however, its exact role in glomerular injury and tubulointerstitial fibrosis is still undefined. The present study was performed to identify the function of NLRP3 in modulating renal injury and fibrosis and the potential involvement of mitochondrial dysfunction in the murine unilateral ureteral obstruction (UUO) model of CKD. Methods. Employing wild-type (WT) and NLRP3-/- mice with or without UUO, we evaluated renal structure, tissue injury, and mitochondrial ultrastructure, as well as expression of some vital molecules involved in the progression of fibrosis, apoptosis, inflammation, and mitochondrial dysfunction. Results. The severe glomerular injury and tubulointerstitial fibrosis induced in WT mice by UUO was markedly attenuated in NLRP3-/- mice as evidenced by blockade of extracellular matrix deposition, decreased cell apoptosis, and phenotypic alterations. Moreover, NLRP3 deletion reversed UUO-induced impairment of mitochondrial morphology and function. Conclusions. NLRP3 deletion ameliorates mitochondrial dysfunction and alleviates renal fibrosis in a murine UUO model of CKD.
Conflict of interest statement
The authors declare that there is no conflict of interests.
Figures




Similar articles
-
H2S alleviates renal injury and fibrosis in response to unilateral ureteral obstruction by regulating macrophage infiltration via inhibition of NLRP3 signaling.Exp Cell Res. 2020 Feb 1;387(1):111779. doi: 10.1016/j.yexcr.2019.111779. Epub 2019 Dec 14. Exp Cell Res. 2020. PMID: 31846625
-
Fluorofenidone attenuates interleukin-1β production by interacting with NLRP3 inflammasome in unilateral ureteral obstruction.Nephrology (Carlton). 2018 Jun;23(6):573-584. doi: 10.1111/nep.13062. Nephrology (Carlton). 2018. PMID: 28437591
-
Inflammasome-Independent Role of NLRP3 Mediates Mitochondrial Regulation in Renal Injury.Front Immunol. 2018 Nov 12;9:2563. doi: 10.3389/fimmu.2018.02563. eCollection 2018. Front Immunol. 2018. PMID: 30483252 Free PMC article.
-
Mitochondrial dysfunction and endoplasmic reticulum stress in the promotion of fibrosis in obstructive nephropathy induced by unilateral ureteral obstruction.Biofactors. 2020 Sep;46(5):716-733. doi: 10.1002/biof.1673. Epub 2020 Sep 9. Biofactors. 2020. PMID: 32905648 Review.
-
DAMPs in Unilateral Ureteral Obstruction.Front Immunol. 2020 Oct 7;11:581300. doi: 10.3389/fimmu.2020.581300. eCollection 2020. Front Immunol. 2020. PMID: 33117389 Free PMC article. Review.
Cited by
-
Astragaloside IV alleviates renal fibrosis by inhibiting renal tubular epithelial cell pyroptosis induced by urotensin II through regulating the cAMP/PKA signaling pathway.PLoS One. 2024 May 31;19(5):e0304365. doi: 10.1371/journal.pone.0304365. eCollection 2024. PLoS One. 2024. PMID: 38820434 Free PMC article.
-
Macrophages in Renal Injury, Repair, Fibrosis Following Acute Kidney Injury and Targeted Therapy.Front Immunol. 2022 Jul 13;13:934299. doi: 10.3389/fimmu.2022.934299. eCollection 2022. Front Immunol. 2022. PMID: 35911736 Free PMC article.
-
The Molecular Mechanism of Renal Tubulointerstitial Inflammation Promoting Diabetic Nephropathy.Int J Nephrol Renovasc Dis. 2023 Dec 5;16:241-252. doi: 10.2147/IJNRD.S436791. eCollection 2023. Int J Nephrol Renovasc Dis. 2023. PMID: 38075191 Free PMC article. Review.
-
Mitochondrial dysfunction/NLRP3 inflammasome axis contributes to angiotensin II-induced skeletal muscle wasting via PPAR-γ.Lab Invest. 2020 May;100(5):712-726. doi: 10.1038/s41374-019-0355-1. Epub 2019 Dec 19. Lab Invest. 2020. PMID: 31857693
-
Chronic kidney disease and NLRP3 inflammasome: Pathogenesis, development and targeted therapeutic strategies.Biochem Biophys Rep. 2022 Dec 26;33:101417. doi: 10.1016/j.bbrep.2022.101417. eCollection 2023 Mar. Biochem Biophys Rep. 2022. PMID: 36620089 Free PMC article. Review.
References
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases

