Azelnidipine, Not Amlodipine, Induces Secretion of Vascular Endothelial Growth Factor From Smooth Muscle Cells and Promotes Endothelial Tube Formation
- PMID: 28348712
- PMCID: PMC5358119
- DOI: 10.14740/cr352w
Azelnidipine, Not Amlodipine, Induces Secretion of Vascular Endothelial Growth Factor From Smooth Muscle Cells and Promotes Endothelial Tube Formation
Abstract
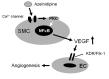
Background: We previously reported that the calcium channel blocker (CCB) nifedipine-induced secretion of vascular endothelial growth factor (VEGF) from human coronary smooth muscle cells (HCSMCs) promoted human coronary endothelial cell (HCEC) tube formation. Therefore, we analyzed whether other CCBs, azelnidipine and amlodipine, also induced the secretion of VEGF and promoted HCEC tube formation, and the underlying molecular mechanisms.
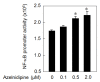
Methods: To evaluate the tube formation, HCECs were grown on Matrigel for 18 hours in the supernatants from HCSMCs that had been treated with different kinds of reagents. Concentrations of VEGF in cultured HCSMCs were determined by specific enzyme immunoassays. Nuclear extracts from HCSMCs were prepared, and nuclear factor-kappa B (NF-κB) activation was measured by EZ-DetectTM Transcription Factor Kits for NF-κB p50 or p65.
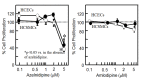
Results: Although azelnidipine dose-dependently stimulated the significant secretion of VEGF from HCSMCs and this stimulation was abolished by a protein kinase C inhibitor, amlodipine-induced secretion of VEGF was significantly lower than that induced by azelnidipine. The medium derived from azelnidipine (at up to 2 μM)-treated HCSMCs led to HCEC tube formation, whereas that obtained with amlodipine did not. Azelnidipine-induced tube formation was blocked by an inhibitor of kinase insert domain-containing receptor/fetal liver kinase-1 tyrosine kinase. Azelnidipine at up to 2 μM induced NF-κB activation.
Conclusions: Azelnidipine, but not amlodipine, stimulated the secretion of VEGF from HCSMCs and induced HCEC tube formation. This secretion is mediated at least in part via the activation of NF-κB. Azelnidipine may have a novel beneficial effect in improving coronary microvascular blood flow in addition to its main effect of lowering blood pressure.
Keywords: Calcium channel blocker; Endothelial cell tube formation; Nuclear factor-kappa B; Smooth muscle cell; Vascular endothelial growth factor.
Figures



Similar articles
-
Nifedipine-induced vascular endothelial growth factor secretion from coronary smooth muscle cells promotes endothelial tube formation via the kinase insert domain-containing receptor/fetal liver kinase-1/NO pathway.Hypertens Res. 2005 Feb;28(2):147-53. doi: 10.1291/hypres.28.147. Hypertens Res. 2005. PMID: 16025742
-
Transactivation of KDR/Flk-1 by the B2 receptor induces tube formation in human coronary endothelial cells.Hypertension. 2003 May;41(5):1118-23. doi: 10.1161/01.HYP.0000064345.33807.57. Epub 2003 Mar 24. Hypertension. 2003. PMID: 12654712
-
Azelnidipine, a new calcium channel blocker, inhibits endothelial inflammatory response by reducing intracellular levels of reactive oxygen species.Eur J Pharmacol. 2006 Sep 28;546(1-3):11-8. doi: 10.1016/j.ejphar.2006.07.030. Epub 2006 Jul 25. Eur J Pharmacol. 2006. PMID: 16919261
-
Chronotropic effects of azelnidipine, a slow- and long-acting dihydropyridine-type calcium channel blocker, in anesthetized dogs: a comparison with amlodipine.J Cardiovasc Pharmacol. 2009 Apr;53(4):325-32. doi: 10.1097/FJC.0b013e31819f1be1. J Cardiovasc Pharmacol. 2009. PMID: 19295444
-
Therapeutic Usefulness of a Novel Calcium Channel Blocker Azelnidipine in the Treatment of Hypertension: A Narrative Review.Cardiol Ther. 2022 Dec;11(4):473-489. doi: 10.1007/s40119-022-00276-4. Epub 2022 Aug 13. Cardiol Ther. 2022. PMID: 35969319 Free PMC article. Review.
Cited by
-
L-type calcium channel blocker increases VEGF concentrations in retinal cells and human serum.PLoS One. 2023 Apr 13;18(4):e0284364. doi: 10.1371/journal.pone.0284364. eCollection 2023. PLoS One. 2023. PMID: 37053203 Free PMC article.
-
Neurolysin substrates bradykinin, neurotensin and substance P enhance brain microvascular permeability in a human in vitro model.J Neuroendocrinol. 2021 Feb;33(2):e12931. doi: 10.1111/jne.12931. Epub 2021 Jan 28. J Neuroendocrinol. 2021. PMID: 33506602 Free PMC article.
References
-
- Cheng XW, Okumura K, Kuzuya M, Jin Z, Nagata K, Obata K, Inoue A. et al. Mechanism of diastolic stiffening of the failing myocardium and its prevention by angiotensin receptor and calcium channel blockers. J Cardiovasc Pharmacol. 2009;54(1):47–56. doi: 10.1097/FJC.0b013e3181ab371d. - DOI - PMC - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
