BAG5 Interacts with DJ-1 and Inhibits the Neuroprotective Effects of DJ-1 to Combat Mitochondrial Oxidative Damage
- PMID: 28348719
- PMCID: PMC5352890
- DOI: 10.1155/2017/5094934
BAG5 Interacts with DJ-1 and Inhibits the Neuroprotective Effects of DJ-1 to Combat Mitochondrial Oxidative Damage
Abstract
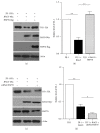
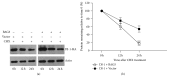
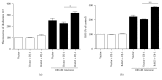
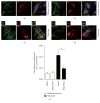
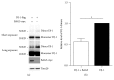
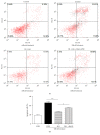
Loss-of-function mutations in gene encoding DJ-1 contribute to the pathogenesis of autosomal recessive early-onset familial forms of Parkinson's disease (PD). DJ-1 is a multifunctional protein and plays a protective role against oxidative stress-induced mitochondrial damage and cell death, but the exact mechanism underlying this is not yet clearly understood. Here, using coimmunoprecipitation (Co-IP) and immunofluorescence methods, we prove that Bcl-2-associated athanogene 5 (BAG5), a BAG family member, interacts with DJ-1 in mammalian cells. Moreover, we show that BAG5 could decrease stability of DJ-1 and weaken its role in mitochondrial protection probably by influencing dimerization in stress condition. Our study reveals the relationship of BAG5 and DJ-1 suggesting a potential role for BAG5 in the pathogenesis of PD through its functional interactions with DJ-1.
Conflict of interest statement
The authors have no actual or potential conflict of interests to report.
Figures

Similar articles
-
RNAi-mediated knockdown of DJ-1 leads to mitochondrial dysfunction via Akt/GSK-3ß and JNK signaling pathways in dopaminergic neuron-like cells.Brain Res Bull. 2019 Mar;146:228-236. doi: 10.1016/j.brainresbull.2019.01.007. Epub 2019 Jan 8. Brain Res Bull. 2019. PMID: 30634017
-
Molecular Mechanism Underlying Hypoxic Preconditioning-Promoted Mitochondrial Translocation of DJ-1 in Hypoxia/Reoxygenation H9c2 Cells.Molecules. 2019 Dec 24;25(1):71. doi: 10.3390/molecules25010071. Molecules. 2019. PMID: 31878239 Free PMC article.
-
DJ-1 regulating PI3K-Nrf2 signaling plays a significant role in bibenzyl compound 20C-mediated neuroprotection against rotenone-induced oxidative insult.Toxicol Lett. 2017 Apr 5;271:74-83. doi: 10.1016/j.toxlet.2017.02.022. Epub 2017 Feb 27. Toxicol Lett. 2017. PMID: 28245986
-
Role of DJ-1 in the mechanism of pathogenesis of Parkinson's disease.J Bioenerg Biomembr. 2019 Jun;51(3):175-188. doi: 10.1007/s10863-019-09798-4. Epub 2019 May 3. J Bioenerg Biomembr. 2019. PMID: 31054074 Free PMC article. Review.
-
Expression of DJ-1 in Neurodegenerative Disorders.Adv Exp Med Biol. 2017;1037:25-43. doi: 10.1007/978-981-10-6583-5_3. Adv Exp Med Biol. 2017. PMID: 29147901 Review.
Cited by
-
Chaperones and Proteostasis: Role in Parkinson's Disease.Diseases. 2020 Jun 22;8(2):24. doi: 10.3390/diseases8020024. Diseases. 2020. PMID: 32580484 Free PMC article. Review.
-
Neuroprotective effects of baicalin and baicalein on the central nervous system and the underlying mechanisms.Heliyon. 2024 Dec 10;11(1):e41002. doi: 10.1016/j.heliyon.2024.e41002. eCollection 2025 Jan 15. Heliyon. 2024. PMID: 39758400 Free PMC article. Review.
-
The effect of cysteine oxidation on DJ-1 cytoprotective function in human alveolar type II cells.Cell Death Dis. 2019 Sep 2;10(9):638. doi: 10.1038/s41419-019-1833-5. Cell Death Dis. 2019. PMID: 31474749 Free PMC article.
-
Role of mitochondrial quality control in neurodegenerative disease progression.Front Cell Neurosci. 2025 May 20;19:1588645. doi: 10.3389/fncel.2025.1588645. eCollection 2025. Front Cell Neurosci. 2025. PMID: 40463912 Free PMC article. Review.
-
Identification of BAG5 as a Potential Biomarker for Parkinson's Disease Patients With R492X PINK1 Mutation.Front Neurosci. 2022 Jul 27;16:903958. doi: 10.3389/fnins.2022.903958. eCollection 2022. Front Neurosci. 2022. PMID: 35968372 Free PMC article.
References
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

