Clinical Activity of the γ-Secretase Inhibitor PF-03084014 in Adults With Desmoid Tumors (Aggressive Fibromatosis)
- PMID: 28350521
- PMCID: PMC5455706
- DOI: 10.1200/JCO.2016.71.1994
Clinical Activity of the γ-Secretase Inhibitor PF-03084014 in Adults With Desmoid Tumors (Aggressive Fibromatosis)
Abstract
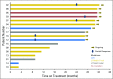
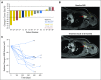
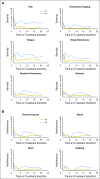
Purpose Desmoid tumors (aggressive fibromatosis) arise from connective tissue cells or fibroblasts. In general, they are slow growing and do not metastasize; however, locally aggressive desmoid tumors can cause severe morbidity and loss of function. Disease recurrence after surgery and/or radiation and diagnosis of multifocal desmoid tumors highlight the need to develop effective systemic treatments for this disease. In this study, we evaluate objective response rate after therapy with the γ-secretase inhibitor PF-03084014 in patients with recurrent, refractory, progressive desmoid tumors. Patients and Methods Seventeen patients with desmoid tumors received PF-03084014 150 mg orally twice a day in 3-week cycles. Response to treatment was evaluated at cycle 1 and every six cycles, that is, 18 weeks, by RECIST (Response Evaluation Criteria in Solid Tumors) version 1.1. Patient-reported outcomes were measured at baseline and at every restaging visit by using the MD Anderson Symptoms Inventory. Archival tumor and blood samples were genotyped for somatic and germline mutations in APC and CTNNB1. Results Of 17 patients accrued to the study, 15 had mutations in APC or CTNNB1 genes. Sixteen patients (94%) were evaluable for response; five (29%) experienced a confirmed partial response and have been on study for more than 2 years. Another five patients with prolonged stable disease as their best response remain on study. Patient-reported outcomes confirmed clinician reporting that the investigational agent was well tolerated and, in subgroup analyses, participants who demonstrated partial response also experienced clinically meaningful and statistically significant improvements in symptom burden. Conclusion PF-03084014 was well tolerated and demonstrated promising clinical benefit in patients with refractory, progressive desmoid tumors who receive long-term treatment.
Figures
Similar articles
-
Safety and efficacy of gamma-secretase inhibitor nirogacestat (PF-03084014) in desmoid tumor: Report of four pediatric/young adult cases.Pediatr Blood Cancer. 2020 Oct;67(10):e28636. doi: 10.1002/pbc.28636. Epub 2020 Aug 6. Pediatr Blood Cancer. 2020. PMID: 32762028
-
Long-Term Follow-Up of Desmoid Fibromatosis Treated with PF-03084014, an Oral Gamma Secretase Inhibitor.Ann Surg Oncol. 2018 Mar;25(3):768-775. doi: 10.1245/s10434-017-6082-1. Epub 2017 Sep 8. Ann Surg Oncol. 2018. PMID: 28887726 Clinical Trial.
-
Nirogacestat, a γ-Secretase Inhibitor for Desmoid Tumors.N Engl J Med. 2023 Mar 9;388(10):898-912. doi: 10.1056/NEJMoa2210140. N Engl J Med. 2023. Update in: Future Oncol. 2025 Apr;21(8):889-901. doi: 10.1080/14796694.2025.2462519. PMID: 36884323 Free PMC article. Updated. Clinical Trial.
-
Examining nirogacestat for adults with progressing desmoid tumors who require systemic treatment.Expert Opin Pharmacother. 2024 Nov;25(16):2115-2124. doi: 10.1080/14656566.2024.2418416. Epub 2024 Oct 18. Expert Opin Pharmacother. 2024. PMID: 39414771 Review.
-
The role of APC and beta-catenin in the aetiology of aggressive fibromatosis (desmoid tumors).Eur J Surg Oncol. 2009 Jan;35(1):3-10. doi: 10.1016/j.ejso.2008.07.003. Epub 2008 Aug 21. Eur J Surg Oncol. 2009. PMID: 18722078 Review.
Cited by
-
Timed Notch Inhibition Drives Photoreceptor Fate Specification in Human Retinal Organoids.Invest Ophthalmol Vis Sci. 2022 Sep 1;63(10):12. doi: 10.1167/iovs.63.10.12. Invest Ophthalmol Vis Sci. 2022. PMID: 36129723 Free PMC article.
-
Informed by Cancer Stem Cells of Solid Tumors: Advances in Treatments Targeting Tumor-Promoting Factors and Pathways.Int J Mol Sci. 2024 Apr 7;25(7):4102. doi: 10.3390/ijms25074102. Int J Mol Sci. 2024. PMID: 38612911 Free PMC article. Review.
-
Locally Aggressive Connective Tissue Tumors.J Clin Oncol. 2018 Jan 10;36(2):202-209. doi: 10.1200/JCO.2017.75.8482. Epub 2017 Dec 8. J Clin Oncol. 2018. PMID: 29220303 Free PMC article. Review.
-
Nirogacestat-the pathway to approval of the first treatment for desmoid tumors, a rare disease.Ther Adv Rare Dis. 2025 Feb 11;6:26330040251317546. doi: 10.1177/26330040251317546. eCollection 2025 Jan-Dec. Ther Adv Rare Dis. 2025. PMID: 39944188 Free PMC article. Review.
-
Wnt Signaling and Its Significance Within the Tumor Microenvironment: Novel Therapeutic Insights.Front Immunol. 2019 Dec 16;10:2872. doi: 10.3389/fimmu.2019.02872. eCollection 2019. Front Immunol. 2019. PMID: 31921137 Free PMC article. Review.
References
-
- de Bree E, Keus R, Melissas J, et al. Desmoid tumors: Need for an individualized approach. Expert Rev Anticancer Ther. 2009;9:525–535. - PubMed
-
- Lev D, Kotilingam D, Wei C, et al. Optimizing treatment of desmoid tumors. J Clin Oncol. 2007;25:1785–1791. - PubMed
-
- Lazar AJF, Hajibashi S, Lev D. Desmoid tumor: From surgical extirpation to molecular dissection. Curr Opin Oncol. 2009;21:352–359. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

