Management of metastatic castration-resistant prostate cancer: Insights from urology experts in Thailand
- PMID: 28352616
- PMCID: PMC5357973
- DOI: 10.1016/j.prnil.2017.01.002
Management of metastatic castration-resistant prostate cancer: Insights from urology experts in Thailand
Abstract
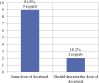
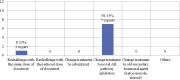
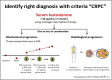
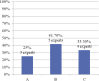
Treatment options for castration-resistant prostate cancer (CRPC) are available, but clear instructions for the selection of appropriate treatment are lacking. A meeting of urology experts based in Thailand was convened with the following objectives: (1) to reach a consensus and share real-life experiences about how to identify CRPC; (2) to choose the appropriate treatment for CRPC patients; (3) to evaluate disease progression using novel inhibitors of the androgen receptor pathway; (4) to identify the frequency of monitoring disease; and (5) to promote rational use of corticosteroids in CRPC patients. This consensus document can provide guidance to other urologists in Thailand to provide appropriate treatment to metastatic CRPC patients in a timely manner.
Keywords: Abiraterone acetate; Androgen receptor; Metastatic castration-resistant prostate cancer; Prostate-specific antigen; Urology expert.
Figures







Similar articles
-
EAU-ESTRO-SIOG Guidelines on Prostate Cancer. Part II: Treatment of Relapsing, Metastatic, and Castration-Resistant Prostate Cancer.Eur Urol. 2017 Apr;71(4):630-642. doi: 10.1016/j.eururo.2016.08.002. Epub 2016 Aug 31. Eur Urol. 2017. PMID: 27591931
-
Consensus on diagnosis and management of non-metastatic castration resistant prostate cancer in Brazil: focus on patient, selection, treatment efficacy, side effects and physician's perception according to patient comorbidities.Int Braz J Urol. 2021 Mar-Apr;47(2):359-373. doi: 10.1590/S1677-5538.IBJU.2020.0249. Int Braz J Urol. 2021. PMID: 33284538 Free PMC article.
-
Current management of advanced and castration resistant prostate cancer.Can J Urol. 2014 Apr;21(2 Supp 1):1-6. Can J Urol. 2014. PMID: 24775717
-
Mechanisms of resistance in castration-resistant prostate cancer (CRPC).Transl Androl Urol. 2015 Jun;4(3):365-80. doi: 10.3978/j.issn.2223-4683.2015.05.02. Transl Androl Urol. 2015. PMID: 26814148 Free PMC article. Review.
-
Abiraterone in the treatment of metastatic castration-resistant prostate cancer.Cancer Manag Res. 2014 Jan 28;6:39-51. doi: 10.2147/CMAR.S39318. eCollection 2014. Cancer Manag Res. 2014. PMID: 24501545 Free PMC article. Review.
Cited by
-
Optimizing outcomes for patients with metastatic prostate cancer: insights from South East Asia Expert Panel.Ther Adv Med Oncol. 2021 Feb 23;13:1758835920985464. doi: 10.1177/1758835920985464. eCollection 2021. Ther Adv Med Oncol. 2021. PMID: 33747148 Free PMC article. Review.
References
-
- James N.D., Sydes M.R., Clarke N.W., Mason M.D., Dearnaley D.P., Spears M.R. Addition of docetaxel, zoledronic acid, or both to first-line long-term hormone therapy in prostate cancer (STAMPEDE): survival results from an adaptive, multiarm, multistage, platform randomised controlled trial. Lancet. 2016;387:1163–1177. - PMC - PubMed
-
- European Association of Urology guideline version 2015 [Internet]. Available from: http://www.uroweb.org/guidelines/.
-
- Ryan C.J., Smith M.R., Fizazi K., Saad F., Mulders P.F.A., Sternberg C.N. Abiraterone acetate plus prednisone versus placebo plus prednisone in chemotherapy-naive men with metastatic castration-resistant prostate cancer (COU-AA-302): final overall survival analysis of a randomised, double-blind, placebo-controlled phase 3 study. Lancet Oncol. 2015;16:152–160. - PubMed
Publication types
LinkOut - more resources
Full Text Sources
Other Literature Sources
