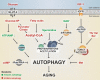
Metabolites in aging and autophagy
- PMID: 28357231
- PMCID: PMC5349198
- DOI: 10.15698/mic2014.04.142
Metabolites in aging and autophagy
Keywords: acetyl-CoA; aging; amino acids; autophagy; metabolism; polyamines.
Conflict of interest statement
Conflict of interest: The authors declare no conflict of interest.
Figures
Similar articles
-
Acetyl-CoA carboxylase 1-dependent lipogenesis promotes autophagy downstream of AMPK.J Biol Chem. 2019 Aug 9;294(32):12020-12039. doi: 10.1074/jbc.RA118.007020. Epub 2019 Jun 17. J Biol Chem. 2019. PMID: 31209110 Free PMC article.
-
Acetyl-CoA Metabolism and Histone Acetylation in the Regulation of Aging and Lifespan.Antioxidants (Basel). 2021 Apr 8;10(4):572. doi: 10.3390/antiox10040572. Antioxidants (Basel). 2021. PMID: 33917812 Free PMC article. Review.
-
Acetyl-CoA synthetase 2 alleviates brain injury following cardiac arrest by promoting autophagy in brain microvascular endothelial cells.Cell Mol Life Sci. 2025 Apr 17;82(1):160. doi: 10.1007/s00018-025-05689-7. Cell Mol Life Sci. 2025. PMID: 40244361 Free PMC article.
-
Metabolomic analyses reveal that anti-aging metabolites are depleted by palmitate but increased by oleate in vivo.Cell Cycle. 2015 Aug 3;14(15):2399-407. doi: 10.1080/15384101.2015.1064206. Epub 2015 Jun 22. Cell Cycle. 2015. PMID: 26098646 Free PMC article.
-
Molecular basis of the 'anti-aging' effect of spermidine and other natural polyamines - a mini-review.Gerontology. 2014;60(4):319-26. doi: 10.1159/000356748. Epub 2014 Jan 28. Gerontology. 2014. PMID: 24481223 Review.
Cited by
-
Acetyl-CoA carboxylase 1-dependent lipogenesis promotes autophagy downstream of AMPK.J Biol Chem. 2019 Aug 9;294(32):12020-12039. doi: 10.1074/jbc.RA118.007020. Epub 2019 Jun 17. J Biol Chem. 2019. PMID: 31209110 Free PMC article.
-
Influence of Normal Aging on Brain Autophagy: A Complex Scenario.Front Aging Neurosci. 2019 Mar 11;11:49. doi: 10.3389/fnagi.2019.00049. eCollection 2019. Front Aging Neurosci. 2019. PMID: 30914945 Free PMC article. Review.
-
Guidelines and recommendations on yeast cell death nomenclature.Microb Cell. 2018 Jan 1;5(1):4-31. doi: 10.15698/mic2018.01.607. Microb Cell. 2018. PMID: 29354647 Free PMC article. Review.
-
Non-genetic impact factors on chronological lifespan and stress resistance of baker's yeast.Microb Cell. 2016 Apr 12;3(6):232-235. doi: 10.15698/mic2016.06.504. Microb Cell. 2016. PMID: 28362008 Free PMC article.
-
Low-Basicity 5-HT6 Receptor Ligands from the Group of Cyclic Arylguanidine Derivatives and Their Antiproliferative Activity Evaluation.Int J Mol Sci. 2024 Sep 24;25(19):10287. doi: 10.3390/ijms251910287. Int J Mol Sci. 2024. PMID: 39408617 Free PMC article.
References
-
- Simonsen A, Cumming RC, Brech A, Isakson P, Schubert DR, Finley KD. Promoting basal levels of autophagy in the nervous system enhances longevity and oxidant resistance in adult Drosophila. Trends Genet TIG. 2008;4(2):176–184. - PubMed
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
