Focal adhesion kinase signaling determines the fate of lung epithelial cells in response to TGF-β
- PMID: 28360109
- PMCID: PMC5495945
- DOI: 10.1152/ajplung.00121.2016
Focal adhesion kinase signaling determines the fate of lung epithelial cells in response to TGF-β
Abstract
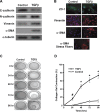
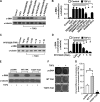
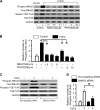
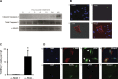
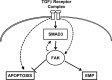
Alveolar epithelial cell (AEC) injury and apoptosis are prominent pathological features of idiopathic pulmonary fibrosis (IPF). There is evidence of AEC plasticity in lung injury repair response and in IPF. In this report, we explore the role of focal adhesion kinase (FAK) signaling in determining the fate of lung epithelial cells in response to transforming growth factor-β1 (TGF-β1). Rat type II alveolar epithelial cells (RLE-6TN) were treated with or without TGF-β1, and the expressions of mesenchymal markers, phenotype, and function were analyzed. Pharmacological protein kinase inhibitors were utilized to screen for SMAD-dependent and -independent pathways. SMAD and FAK signaling was analyzed using siRNA knockdown, inhibitors, and expression of a mutant construct of FAK. Apoptosis was measured using cleaved caspase-3 and terminal deoxynucleotidyl transferase dUTP nick end labeling (TUNEL) staining. TGF-β1 induced the acquisition of mesenchymal markers, including α-smooth muscle actin, in RLE-6TN cells and enhanced the contraction of three-dimensional collagen gels. This phenotypical transition or plasticity, epithelial-myofibroblast plasticity (EMP), is dependent on SMAD3 and FAK signaling. FAK activation was found to be dependent on ALK5/SMAD3 signaling. We observed that TGF-β1 induces both EMP and apoptosis in the same cell culture system but not in the same cell. While blockade of SMAD signaling inhibited EMP, it had a minimal effect on apoptosis; in contrast, inhibition of FAK signaling markedly shifted to an apoptotic fate. The data support that FAK activation determines whether AECs undergo EMP vs. apoptosis in response to TGF-β1 stimulation. TGF-β1-induced EMP is FAK- dependent, whereas TGF-β1-induced apoptosis is favored when FAK signaling is inhibited.
Keywords: FAK; TGF-β1; alveolar epithelial cell; apoptosis; differentiation.
Copyright © 2017 the American Physiological Society.
Figures


References
-
- Cai GQ, Chou CF, Hu M, Zheng A, Reichardt LF, Guan JL, Fang H, Luckhardt TR, Zhou Y, Thannickal VJ, Ding Q. Neuronal Wiskott-Aldrich syndrome protein (N-WASP) is critical for formation of α-smooth muscle actin filaments during myofibroblast differentiation. Am J Physiol Lung Cell Mol Physiol 303: L692–L702, 2012. doi:10.1152/ajplung.00390.2011. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous

