Enhancement of Zika virus pathogenesis by preexisting antiflavivirus immunity
- PMID: 28360135
- PMCID: PMC5714274
- DOI: 10.1126/science.aal4365
Enhancement of Zika virus pathogenesis by preexisting antiflavivirus immunity
Abstract
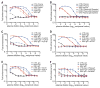
Zika virus (ZIKV) is spreading rapidly into regions around the world where other flaviviruses, such as dengue virus (DENV) and West Nile virus (WNV), are endemic. Antibody-dependent enhancement has been implicated in more severe forms of flavivirus disease, but whether this also applies to ZIKV infection is unclear. Using convalescent plasma from DENV- and WNV-infected individuals, we found substantial enhancement of ZIKV infection in vitro that was mediated through immunoglobulin G engagement of Fcγ receptors. Administration of DENV- or WNV-convalescent plasma into ZIKV-susceptible mice resulted in increased morbidity-including fever, viremia, and viral loads in spinal cord and testes-and increased mortality. Antibody-dependent enhancement may explain the severe disease manifestations associated with recent ZIKV outbreaks and highlights the need to exert great caution when designing flavivirus vaccines.
Copyright © 2017, American Association for the Advancement of Science.
Figures
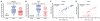
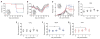
Comment in
-
Dengue Antibodies, then Zika: A Fatal Sequence in Mice.Immunity. 2017 May 16;46(5):771-773. doi: 10.1016/j.immuni.2017.04.023. Immunity. 2017. PMID: 28514683
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

