Killers creating new life: caspases drive apoptosis-induced proliferation in tissue repair and disease
- PMID: 28362431
- PMCID: PMC5520457
- DOI: 10.1038/cdd.2017.47
Killers creating new life: caspases drive apoptosis-induced proliferation in tissue repair and disease
Abstract
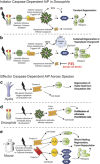
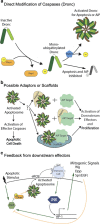
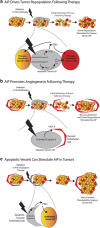
Apoptosis is a carefully orchestrated and tightly controlled form of cell death, conserved across metazoans. As the executioners of apoptotic cell death, cysteine-dependent aspartate-directed proteases (caspases) are critical drivers of this cellular disassembly. Early studies of genetically programmed cell death demonstrated that the selective activation of caspases induces apoptosis and the precise elimination of excess cells, thereby sculpting structures and refining tissues. However, over the past decade there has been a fundamental shift in our understanding of the roles of caspases during cell death-a shift precipitated by the revelation that apoptotic cells actively engage with their surrounding environment throughout the death process, and caspases can trigger a myriad of signals, some of which drive concurrent cell proliferation regenerating damaged structures and building up lost tissues. This caspase-driven compensatory proliferation is referred to as apoptosis-induced proliferation (AiP). Diverse mechanisms of AiP have been found across species, ranging from planaria to mammals. In this review, we summarize the current knowledge of AiP and we highlight recent advances in the field including the involvement of reactive oxygen species and macrophage-like immune cells in one form of AiP, novel regulatory mechanisms affecting caspases during AiP, and emerging clinical data demonstrating the critical importance of AiP in cancer.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

References
-
- Haynie JL, Bryant PJ. The effects of X-rays on the proliferation dynamics of cells in the imaginal wing disc of Drosophila melanogaster. Dev Genes Evol 1977; 183: 85–100. - PubMed
-
- James AA, Bryant PJ. A quantitative study of cell death and mitotic inhibition in gamma-irradiated imaginal wing discs of Drosophila melanogaster. Radiat Res 1981; 87: 552–564. - PubMed
-
- Hwang JS, Kobayashi C, Agata K, Ikeo K, Gojobori T. Detection of apoptosis during planarian regeneration by the expression of apoptosis-related genes and TUNEL assay. Gene 2004; 333: 15–25. - PubMed
-
- Vlaskalin T, Wong CJ, Tsilfidis C. Growth and apoptosis during larval forelimb development and adult forelimb regeneration in the newt (Notophthalmus viridescens. Dev Genes Evol 2004; 214: 423–431. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous

