Complex Routes of Nosocomial Vancomycin-Resistant Enterococcus faecium Transmission Revealed by Genome Sequencing
- PMID: 28362945
- PMCID: PMC5439346
- DOI: 10.1093/cid/ciw872
Complex Routes of Nosocomial Vancomycin-Resistant Enterococcus faecium Transmission Revealed by Genome Sequencing
Abstract
Background: Vancomycin-resistant Enterococcus faecium (VREfm) is a leading cause of nosocomial infection. Here, we describe the utility of whole-genome sequencing in defining nosocomial VREfm transmission.
Methods: A retrospective study at a single hospital in the United Kingdom identified 342 patients with E. faecium bloodstream infection over 7 years. Of these, 293 patients had a stored isolate and formed the basis for the study. The first stored isolate from each case was sequenced (200 VREfm [197 vanA, 2 vanB, and 1 isolate containing both vanA and vanB], 93 vancomycin-susceptible E. faecium) and epidemiological data were collected. Genomes were also available for E. faecium associated with bloodstream infections in 15 patients in neighboring hospitals, and 456 patients across the United Kingdom and Ireland.
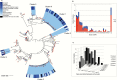
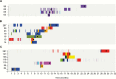
Results: The majority of infections in the 293 patients were hospital-acquired (n = 249) or healthcare-associated (n = 42). Phylogenetic analysis showed that 291 of 293 isolates resided in a hospital-associated clade that contained numerous discrete clusters of closely related isolates, indicative of multiple introductions into the hospital followed by clonal expansion associated with transmission. Fine-scale analysis of 6 exemplar phylogenetic clusters containing isolates from 93 patients (32%) identified complex transmission routes that spanned numerous wards and years, extending beyond the detection of conventional infection control. These contained both vancomycin-resistant and -susceptible isolates. We also identified closely related isolates from patients at Cambridge University Hospitals NHS Foundation Trust and regional and national hospitals, suggesting interhospital transmission.
Conclusions: These findings provide important insights for infection control practice and signpost areas for interventions. We conclude that sequencing represents a powerful tool for the enhanced surveillance and control of nosocomial E. faecium transmission and infection.
Keywords: Enterococcus faecium; genome sequencing.; infection control; transmission; vancomycin resistant.
© The Author 2017. Published by Oxford University Press for the Infectious Diseases Society of America.
Figures

Similar articles
-
Protracted transmission and persistence of ST80 vancomycin-resistant Enterococcus faecium clonal complex types CT2933, CT2932 and CT1916 in a large Irish hospital: a 39-month whole-genome sequencing study.J Hosp Infect. 2024 Sep;151:11-20. doi: 10.1016/j.jhin.2024.06.002. Epub 2024 Jun 27. J Hosp Infect. 2024. PMID: 38944282
-
Genomic Surveillance of Enterococcus faecium Reveals Limited Sharing of Strains and Resistance Genes between Livestock and Humans in the United Kingdom.mBio. 2018 Nov 6;9(6):e01780-18. doi: 10.1128/mBio.01780-18. mBio. 2018. PMID: 30401778 Free PMC article.
-
Characterization of vanA-harboring plasmids supports differentiation of outbreak-related and sporadic vancomycin-resistant Enterococcus faecium isolates in a tertiary care hospital.BMC Microbiol. 2025 May 28;25(1):337. doi: 10.1186/s12866-025-04058-5. BMC Microbiol. 2025. PMID: 40437365 Free PMC article.
-
Genomics of vancomycin-resistant Enterococcus faecium.Microb Genom. 2019 Jul;5(7):e000283. doi: 10.1099/mgen.0.000283. Epub 2019 Jul 22. Microb Genom. 2019. PMID: 31329096 Free PMC article. Review.
-
A molecular study regarding the spread of vanA vancomycin-resistant Enterococcus faecium in a tertiary hospital in China.J Glob Antimicrob Resist. 2022 Dec;31:270-278. doi: 10.1016/j.jgar.2022.10.010. Epub 2022 Oct 20. J Glob Antimicrob Resist. 2022. PMID: 36273808 Review.
Cited by
-
The global dissemination of hospital clones of Enterococcus faecium.Genome Med. 2021 Mar 30;13(1):52. doi: 10.1186/s13073-021-00868-0. Genome Med. 2021. PMID: 33785076 Free PMC article.
-
Top-Down Genomic Surveillance Approach To Investigate the Genomic Epidemiology and Antibiotic Resistance Patterns of Enterococcus faecium Detected in Cancer Patients in Arkansas.Microbiol Spectr. 2023 Jun 15;11(3):e0490122. doi: 10.1128/spectrum.04901-22. Epub 2023 Mar 30. Microbiol Spectr. 2023. PMID: 36995227 Free PMC article.
-
Mode and dynamics of vanA-type vancomycin resistance dissemination in Dutch hospitals.Genome Med. 2021 Jan 20;13(1):9. doi: 10.1186/s13073-020-00825-3. Genome Med. 2021. PMID: 33472670 Free PMC article.
-
Co-carriage of diverse vancomycin-resistant Enterococcus faecium ST80-lineages by 70% of patients in an Irish hospital.JAC Antimicrob Resist. 2025 Apr 29;7(3):dlaf065. doi: 10.1093/jacamr/dlaf065. eCollection 2025 Jun. JAC Antimicrob Resist. 2025. PMID: 40309497 Free PMC article.
-
Whole-genome sequencing revealed independent emergence of vancomycin-resistant Enterococcus faecium causing sequential outbreaks over 3 years in a tertiary care hospital.Eur J Clin Microbiol Infect Dis. 2019 Jun;38(6):1163-1170. doi: 10.1007/s10096-019-03524-z. Epub 2019 Mar 19. Eur J Clin Microbiol Infect Dis. 2019. PMID: 30888549
References
-
- Sievert DM, Ricks P, Edwards JR, et al. Antimicrobial-resistant pathogens associated with healthcare-associated infections: summary of data reported to the National Healthcare Safety Network at the Centers for Disease Control and Prevention, 2009–2010. Infect Control Hosp Epidemiol 2013; 34:1–14. - PubMed
-
- Leclercq R, Derlot E, Duval J, Courvalin P. Plasmid-mediated resistance to vancomycin and teicoplanin in Enterococcus faecium. N Engl J Med 1988; 319:157–61. - PubMed
-
- Uttley AH, Collins CH, Naidoo J, George RC. Vancomycin-resistant enterococci. Lancet 1988; 1:57–8. - PubMed
-
- Cheah AL, Spelman T, Liew D, et al. Enterococcal bacteraemia: factors influencing mortality, length of stay and costs of hospitalization. Clin Microbiol Infect 2013; 19:E181–9. - PubMed
-
- Stosor V, Peterson LR, Postelnick M, Noskin GA. Enterococcus faecium bacteremia: does vancomycin resistance make a difference? Arch Intern Med 1998; 158:522–7. - PubMed
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

