Toward understanding the role of cartilage particulates in synovial inflammation
- PMID: 28365462
- PMCID: PMC5554538
- DOI: 10.1016/j.joca.2017.03.015
Toward understanding the role of cartilage particulates in synovial inflammation
Abstract
Objective: Arthroscopy with lavage and synovectomy can remove tissue debris from the joint space and the synovial lining to provide pain relief to patients with osteoarthritis (OA). Here, we developed an in vitro model to study the interaction of cartilage wear particles with fibroblast-like synoviocytes (FLS) to better understand the interplay of cartilage particulates with cytokines on cells of the synovium.
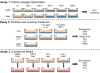
Method: In this study sub-10 μm cartilage particles or 1 μm latex particles were co-cultured with FLS ±10 ng/mL interleukin-1α (IL-1α) or tumor necrosis factor-α (TNF-α). Samples were analyzed for DNA, glycosaminoglycan (GAG), and collagen, and media samples were analyzed for media GAG, nitric oxide (NO) and prostaglandin-E2 (PGE2). The nature of the physical interaction between the particles and FLS was determined by microscopy.
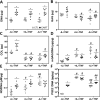
Results: Both latex and cartilage particles could be phagocytosed by FLS. Cartilage particles were internalized and attached to the surface of both dense monolayers and individual cells. Co-culture of FLS with cartilage particulates resulted in a significant increase in cell sheet DNA and collagen content as well as NO and PGE2 synthesis compared to control and latex treated groups.
Conclusion: The proliferative response of FLS to cartilage wear particles resulted in an overall increase in extracellular matrix (ECM) content, analogous to the thickening of the synovial lining observed in OA patients. Understanding how cartilage particles interface with the synovium may provide insight into how this interaction contributes to OA progression and may guide the role of lavage and synovectomy for degenerative disease.
Keywords: Cartilage; Inflammation; Synovium; Wear particles.
Copyright © 2017 Osteoarthritis Research Society International. Published by Elsevier Ltd. All rights reserved.
Conflict of interest statement
None of authors had a financial or personal relationship with organizations that could influence the work presented in this manuscript.
Figures





Similar articles
-
Cadherin-11 involves in synovitis and increases the migratory and invasive capacity of fibroblast-like synoviocytes of osteoarthritis.Int Immunopharmacol. 2015 May;26(1):153-61. doi: 10.1016/j.intimp.2015.03.024. Epub 2015 Mar 28. Int Immunopharmacol. 2015. PMID: 25824718
-
The emerging role of fibroblast-like synoviocytes-mediated synovitis in osteoarthritis: An update.J Cell Mol Med. 2020 Sep;24(17):9518-9532. doi: 10.1111/jcmm.15669. Epub 2020 Jul 19. J Cell Mol Med. 2020. PMID: 32686306 Free PMC article. Review.
-
Upregulated expression of CCR3 in osteoarthritis and CCR3 mediated activation of fibroblast-like synoviocytes.Cytokine. 2016 Jan;77:211-9. doi: 10.1016/j.cyto.2015.09.012. Epub 2015 Sep 26. Cytokine. 2016. PMID: 26409848
-
The role of cytokines in osteoarthritis pathophysiology.Biorheology. 2002;39(1-2):237-46. Biorheology. 2002. PMID: 12082286 Review.
-
Polyethylene wear particles play a role in development of osteoarthritis via detrimental effects on cartilage, meniscus, and synovium.Osteoarthritis Cartilage. 2013 Dec;21(12):2021-9. doi: 10.1016/j.joca.2013.09.013. Epub 2013 Oct 23. Osteoarthritis Cartilage. 2013. PMID: 24161707
Cited by
-
Overcoming barriers for intra-articular delivery of disease-modifying osteoarthritis drugs.Trends Pharmacol Sci. 2022 Mar;43(3):171-187. doi: 10.1016/j.tips.2021.12.004. Epub 2022 Jan 24. Trends Pharmacol Sci. 2022. PMID: 35086691 Free PMC article. Review.
-
Role of crosstalk between synovial cells and chondrocytes in osteoarthritis (Review).Exp Ther Med. 2024 Mar 15;27(5):201. doi: 10.3892/etm.2024.12490. eCollection 2024 May. Exp Ther Med. 2024. PMID: 38590580 Free PMC article. Review.
-
Sustained low-dose dexamethasone delivery via a PLGA microsphere-embedded agarose implant for enhanced osteochondral repair.Acta Biomater. 2020 Jan 15;102:326-340. doi: 10.1016/j.actbio.2019.11.052. Epub 2019 Dec 2. Acta Biomater. 2020. PMID: 31805408 Free PMC article.
-
Integrin signalling in joint development, homeostasis and osteoarthritis.Nat Rev Rheumatol. 2024 Aug;20(8):492-509. doi: 10.1038/s41584-024-01130-8. Epub 2024 Jul 16. Nat Rev Rheumatol. 2024. PMID: 39014254 Free PMC article. Review.
-
Paeoniflorin, the Main Monomer Component of Paeonia lactiflora, Exhibits Anti-inflammatory Properties in Osteoarthritis Synovial Inflammation.Chin J Integr Med. 2024 May;30(5):433-442. doi: 10.1007/s11655-023-3653-9. Epub 2023 Nov 24. Chin J Integr Med. 2024. PMID: 37999887
References
-
- Griffin TM, Guilak F. The role of mechanical loading in the onset and progression of osteoarthritis. Exerc Sport Sci Rev. 2005;33:195–200. - PubMed
-
- Kuster MS, Podsiadlo P, Stachowiak GW. Shape of wear particles found in the human knee joint and their relationship to osteoarthritis. Rheumatology. 1998:978–984. - PubMed
-
- Hakshur K, Benhar I, Bar-Ziv Y, Halperin N, Segal D, Eliaz N. Acta Biomaterialia. Acta Materialia Inc; 2011. The effect of hyaluronan injections into human knees on the number of bone and cartilage wear particles captured by bio-ferrography; pp. 848–857. - PubMed
-
- Evans CH, Mears DC, Mcknight JL. A preliminary ferrographic survey of the wear particles in human synovial fluid. Arthritis & Rheumatism. 1981:912–918. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

