RIPK3 Restricts Viral Pathogenesis via Cell Death-Independent Neuroinflammation
- PMID: 28366204
- PMCID: PMC5405738
- DOI: 10.1016/j.cell.2017.03.011
RIPK3 Restricts Viral Pathogenesis via Cell Death-Independent Neuroinflammation
Abstract
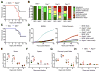
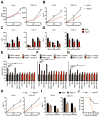
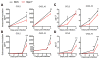
Receptor-interacting protein kinase-3 (RIPK3) is an activator of necroptotic cell death, but recent work has implicated additional roles for RIPK3 in inflammatory signaling independent of cell death. However, while necroptosis has been shown to contribute to antiviral immunity, death-independent roles for RIPK3 in host defense have not been demonstrated. Using a mouse model of West Nile virus (WNV) encephalitis, we show that RIPK3 restricts WNV pathogenesis independently of cell death. Ripk3-/- mice exhibited enhanced mortality compared to wild-type (WT) controls, while mice lacking the necroptotic effector MLKL, or both MLKL and caspase-8, were unaffected. The enhanced susceptibility of Ripk3-/- mice arose from suppressed neuronal chemokine expression and decreased central nervous system (CNS) recruitment of T lymphocytes and inflammatory myeloid cells, while peripheral immunity remained intact. These data identify pleiotropic functions for RIPK3 in the restriction of viral pathogenesis and implicate RIPK3 as a key coordinator of immune responses within the CNS.
Keywords: RIPK1; RIPK3; West Nile virus; chemokines; necroptosis; neuroimmunology; neuroinflammation.
Copyright © 2017 Elsevier Inc. All rights reserved.
Figures
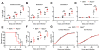


Comment in
-
A RIPtide Protects Neurons from Infection.Cell Host Microbe. 2017 Apr 12;21(4):415-416. doi: 10.1016/j.chom.2017.03.014. Cell Host Microbe. 2017. PMID: 28407477
-
RIPped for neuroinflammation.Cell Res. 2017 Sep;27(9):1081-1082. doi: 10.1038/cr.2017.75. Epub 2017 May 19. Cell Res. 2017. PMID: 28524164 Free PMC article.
-
Host response: Neurons loosen the gRIP of death.Nat Microbiol. 2017 May 22;2:17090. doi: 10.1038/nmicrobiol.2017.90. Nat Microbiol. 2017. PMID: 28530667 No abstract available.
References
-
- Beisner DR, Ch’en IL, Kolla RV, Hoffmann A, Hedrick SM. Cutting edge: innate immunity conferred by B cells is regulated by caspase-8. J Immunol. 2005;175:3469–3473. - PubMed
-
- Berger SB, Kasparcova V, Hoffman S, Swift B, Dare L, Schaeffer M, Capriotti C, Cook M, Finger J, Hughes-Earle A, et al. Cutting Edge: RIP1 kinase activity is dispensable for normal development but is a key regulator of inflammation in SHARPIN-deficient mice. J Immunol. 2014;192:5476–5480. - PMC - PubMed
-
- Brien JD, Lazear HM, Diamond MS. Propagation, quantification, detection, and storage of West Nile virus. Current protocols in microbiology. 2013;31:15D 13 11–15D 13 18. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Miscellaneous

