Major Histocompatibility Complex (MHC) Class I and MHC Class II Proteins: Conformational Plasticity in Antigen Presentation
- PMID: 28367149
- PMCID: PMC5355494
- DOI: 10.3389/fimmu.2017.00292
Major Histocompatibility Complex (MHC) Class I and MHC Class II Proteins: Conformational Plasticity in Antigen Presentation
Abstract
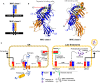
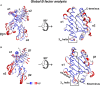
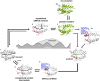
Antigen presentation by major histocompatibility complex (MHC) proteins is essential for adaptive immunity. Prior to presentation, peptides need to be generated from proteins that are either produced by the cell's own translational machinery or that are funneled into the endo-lysosomal vesicular system. The prolonged interaction between a T cell receptor and specific pMHC complexes, after an extensive search process in secondary lymphatic organs, eventually triggers T cells to proliferate and to mount a specific cellular immune response. Once processed, the peptide repertoire presented by MHC proteins largely depends on structural features of the binding groove of each particular MHC allelic variant. Additionally, two peptide editors-tapasin for class I and HLA-DM for class II-contribute to the shaping of the presented peptidome by favoring the binding of high-affinity antigens. Although there is a vast amount of biochemical and structural information, the mechanism of the catalyzed peptide exchange for MHC class I and class II proteins still remains controversial, and it is not well understood why certain MHC allelic variants are more susceptible to peptide editing than others. Recent studies predict a high impact of protein intermediate states on MHC allele-specific peptide presentation, which implies a profound influence of MHC dynamics on the phenomenon of immunodominance and the development of autoimmune diseases. Here, we review the recent literature that describe MHC class I and II dynamics from a theoretical and experimental point of view and we highlight the similarities between MHC class I and class II dynamics despite the distinct functions they fulfill in adaptive immunity.
Keywords: HLA; HLA-DM; adaptive immunity; antigen presentation; major histocompatibility complex; peptide exchange; protein dynamics; tapasin.
Figures


References
Publication types
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

