Comparing multifocal pupillographic objective perimetry (mfPOP) and multifocal visual evoked potentials (mfVEP) in retinal diseases
- PMID: 28368051
- PMCID: PMC5377468
- DOI: 10.1038/srep45847
Comparing multifocal pupillographic objective perimetry (mfPOP) and multifocal visual evoked potentials (mfVEP) in retinal diseases
Abstract
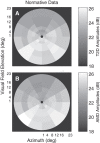
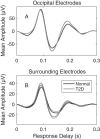
Multifocal pupillographic objective perimetry (mfPOP) shows regions of slight hypersensitivity away from retinal regions damaged by diabetes or age-related macular degeneration (AMD). This study examines if such results also appear in multifocal visual evoked potentials (mfVEPs) recorded on the same day in the same patients. The pupil control system receives input from the extra-striate cortex, so we also examined evidence for such input. We recruited subjects with early type 2 diabetes (T2D) with no retinopathy, and patients with unilateral exudative AMD. Population average responses of the diabetes patients, and the normal fellow eyes of AMD patients, showed multiple regions of significant hypersensitivity (p < 0.05) on both mfPOP and mfVEPs. For mfVEPs the occipital electrodes showed fewer hypersensitive regions than the surrounding electrodes. More advanced AMD showed regions of suppression becoming centrally concentrated in the exudative AMD areas. Thus, mfVEP electrodes biased towards extra-striate cortical responses (surround electrodes) appeared to show similar hypersensitive visual field locations to mfPOP in early stage diabetic and AMD damage. Our findings suggest that hypersensitive regions may be a potential biomarker for future development of AMD or non-proliferative diabetic retinopathy, and may be more informative than visual acuity which remains largely undisturbed during early disease.
Conflict of interest statement
Faran Sabeti – Research support NuCoria Pty Ltd., Acton, ACT. Australia. Andrew James - Research support and Patent NuCoria NuCoria Pty Ltd., Acton, ACT. Australia. Corinne Carle – Patent NuCoria Pty Ltd., Acton, ACT. Australia. Rohan Essex – None. Andrew Bell – None. Ted Maddess - Research support and Patent NuCoria Pty Ltd., Acton, ACT. Australia.
Figures





Similar articles
-
Multifocal pupillographic assessment of age-related macular degeneration.Optom Vis Sci. 2011 Dec;88(12):1477-85. doi: 10.1097/OPX.0b013e318235af61. Optom Vis Sci. 2011. PMID: 21983122
-
Dichoptic multifocal visual evoked potentials identify local retinal dysfunction in age-related macular degeneration.Doc Ophthalmol. 2013 Apr;126(2):125-36. doi: 10.1007/s10633-012-9366-6. Epub 2012 Dec 13. Doc Ophthalmol. 2013. PMID: 23238587
-
Multifocal pupillography identifies retinal dysfunction in early age-related macular degeneration.Graefes Arch Clin Exp Ophthalmol. 2013 Jul;251(7):1707-16. doi: 10.1007/s00417-013-2273-z. Epub 2013 Feb 8. Graefes Arch Clin Exp Ophthalmol. 2013. PMID: 23392820
-
[Multifocal visual electrophysiology in visual function evaluation].Fa Yi Xue Za Zhi. 2013 Aug;29(4):286-9, 294. Fa Yi Xue Za Zhi. 2013. PMID: 24350547 Review. Chinese.
-
The Role of mf-ERG in the Diagnosis and Treatment of Age-Related Macular Degeneration: Electrophysiological Features of AMD.Semin Ophthalmol. 2018;33(4):461-469. doi: 10.1080/08820538.2017.1301496. Epub 2017 Mar 22. Semin Ophthalmol. 2018. PMID: 28328288 Review.
Cited by
-
Mapping simulated visual field defects with movie-viewing pupil perimetry.Graefes Arch Clin Exp Ophthalmol. 2025 Jun;263(6):1641-1650. doi: 10.1007/s00417-024-06733-1. Epub 2025 Jan 9. Graefes Arch Clin Exp Ophthalmol. 2025. PMID: 39779511 Free PMC article.
-
Basics, benefits, and pitfalls of pupillometers assessing visual function.Eye (Lond). 2024 Aug;38(12):2415-2421. doi: 10.1038/s41433-024-03151-9. Epub 2024 May 27. Eye (Lond). 2024. PMID: 38802485 Free PMC article. Review.
-
Discriminating early-stage diabetic retinopathy with subjective and objective perimetry.Front Endocrinol (Lausanne). 2024 Jan 8;14:1333826. doi: 10.3389/fendo.2023.1333826. eCollection 2023. Front Endocrinol (Lausanne). 2024. PMID: 38264290 Free PMC article.
-
Recovery dynamics of multifocal pupillographic objective perimetry from tropicamide dilation.Graefes Arch Clin Exp Ophthalmol. 2020 Jan;258(1):191-200. doi: 10.1007/s00417-019-04523-8. Epub 2019 Nov 13. Graefes Arch Clin Exp Ophthalmol. 2020. PMID: 31720837
-
Retinotopic effects of visual attention revealed by dichoptic multifocal pupillography.Sci Rep. 2018 Feb 14;8(1):2991. doi: 10.1038/s41598-018-21196-1. Sci Rep. 2018. PMID: 29445236 Free PMC article.
References
MeSH terms
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

