Memory and synaptic plasticity are impaired by dysregulated hippocampal O-GlcNAcylation
- PMID: 28368052
- PMCID: PMC5377249
- DOI: 10.1038/srep44921
Memory and synaptic plasticity are impaired by dysregulated hippocampal O-GlcNAcylation
Abstract
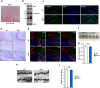
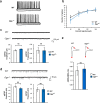
O-GlcNAcylated proteins are abundant in the brain and are associated with neuronal functions and neurodegenerative diseases. Although several studies have reported the effects of aberrant regulation of O-GlcNAcylation on brain function, the roles of O-GlcNAcylation in synaptic function remain unclear. To understand the effect of aberrant O-GlcNAcylation on the brain, we used Oga+/- mice which have an increased level of O-GlcNAcylation, and found that Oga+/- mice exhibited impaired spatial learning and memory. Consistent with this result, Oga+/- mice showed a defect in hippocampal synaptic plasticity. Oga heterozygosity causes impairment of both long-term potentiation and long-term depression due to dysregulation of AMPA receptor phosphorylation. These results demonstrate a role for hyper-O-GlcNAcylation in learning and memory.
Conflict of interest statement
The authors declare no competing financial interests.
Figures



References
-
- Okuyama R. & Marshall S. UDP-N-acetylglucosaminyl transferase (OGT) in brain tissue: temperature sensitivity and subcellular distribution of cytosolic and nuclear enzyme. J Neurochem 86, 1271–1280 (2003). - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

