The hidden life of integrative and conjugative elements
- PMID: 28369623
- PMCID: PMC5812530
- DOI: 10.1093/femsre/fux008
The hidden life of integrative and conjugative elements
Abstract
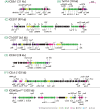
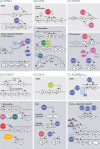
Integrative and conjugative elements (ICEs) are widespread mobile DNA that transmit both vertically, in a host-integrated state, and horizontally, through excision and transfer to new recipients. Different families of ICEs have been discovered with more or less restricted host ranges, which operate by similar mechanisms but differ in regulatory networks, evolutionary origin and the types of variable genes they contribute to the host. Based on reviewing recent experimental data, we propose a general model of ICE life style that explains the transition between vertical and horizontal transmission as a result of a bistable decision in the ICE-host partnership. In the large majority of cells, the ICE remains silent and integrated, but hidden at low to very low frequencies in the population specialized host cells appear in which the ICE starts its process of horizontal transmission. This bistable process leads to host cell differentiation, ICE excision and transfer, when suitable recipients are present. The ratio of ICE bistability (i.e. ratio of horizontal to vertical transmission) is the outcome of a balance between fitness costs imposed by the ICE horizontal transmission process on the host cell, and selection for ICE distribution (i.e. ICE 'fitness'). From this emerges a picture of ICEs as elements that have adapted to a mostly confined life style within their host, but with a very effective and dynamic transfer from a subpopulation of dedicated cells.
Keywords: bistability; cellular differentiation; fitness cost; horizontal gene transfer.
© FEMS 2017.
Figures




References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

