Memory of recent oxygen experience switches pheromone valence in Caenorhabditis elegans
- PMID: 28373553
- PMCID: PMC5402444
- DOI: 10.1073/pnas.1618934114
Memory of recent oxygen experience switches pheromone valence in Caenorhabditis elegans
Abstract
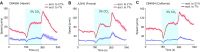
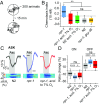
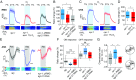
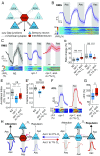
Animals adjust their behavioral priorities according to momentary needs and prior experience. We show that Caenorhabditis elegans changes how it processes sensory information according to the oxygen environment it experienced recently. C. elegans acclimated to 7% O2 are aroused by CO2 and repelled by pheromones that attract animals acclimated to 21% O2 This behavioral plasticity arises from prolonged activity differences in a circuit that continuously signals O2 levels. A sustained change in the activity of O2-sensing neurons reprograms the properties of their postsynaptic partners, the RMG hub interneurons. RMG is gap-junctionally coupled to the ASK and ADL pheromone sensors that respectively drive pheromone attraction and repulsion. Prior O2 experience has opposite effects on the pheromone responsiveness of these neurons. These circuit changes provide a physiological correlate of altered pheromone valence. Our results suggest C. elegans stores a memory of recent O2 experience in the RMG circuit and illustrate how a circuit is flexibly sculpted to guide behavioral decisions in a context-dependent manner.
Keywords: acclimation; experience-dependent plasticity; neural circuit; oxygen sensing; tonic circuit.
Conflict of interest statement
The authors declare no conflict of interest.
Figures


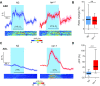
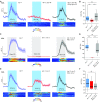
References
-
- Saeki S, Yamamoto M, Iino Y. Plasticity of chemotaxis revealed by paired presentation of a chemoattractant and starvation in the nematode Caenorhabditis elegans. J Exp Biol. 2001;204:1757–1764. - PubMed
-
- Kindt KS, et al. Dopamine mediates context-dependent modulation of sensory plasticity in C. elegans. Neuron. 2007;55:662–676. - PubMed
-
- Hoopfer ED. Neural control of aggression in Drosophila. Curr Opin Neurobiol. 2016;38:109–118. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

