Avelumab, an Anti-Programmed Death-Ligand 1 Antibody, In Patients With Refractory Metastatic Urothelial Carcinoma: Results From a Multicenter, Phase Ib Study
- PMID: 28375787
- PMCID: PMC5493051
- DOI: 10.1200/JCO.2016.71.6795
Avelumab, an Anti-Programmed Death-Ligand 1 Antibody, In Patients With Refractory Metastatic Urothelial Carcinoma: Results From a Multicenter, Phase Ib Study
Abstract
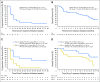
Purpose We assessed the safety and antitumor activity of avelumab, a fully human anti-programmed death-ligand 1 (PD-L1) IgG1 antibody, in patients with refractory metastatic urothelial carcinoma. Methods In this phase Ib, multicenter, expansion cohort, patients with urothelial carcinoma progressing after platinum-based chemotherapy and unselected for PD-L1 expression received avelumab 10 mg/kg intravenously every 2 weeks. The primary objectives were safety and tolerability. Secondary objectives included confirmed objective response rate (Response Evaluation Criteria in Solid Tumors [RECIST] version 1.1), progression-free survival, overall survival (OS), and PD-L1-associated clinical activity. PD-L1 positivity was defined as expression by immunohistochemistry on ≥ 5% of tumor cells. Results Forty-four patients were treated with avelumab and followed for a median of 16.5 months (interquartile range, 15.8 to 16.7 months). The data cutoff was March 19, 2016. The most frequent treatment-related adverse events of any grade were fatigue/asthenia (31.8%), infusion-related reaction (20.5%), and nausea (11.4%). Grades 3 to 4 treatment-related adverse events occurred in three patients (6.8%) and included asthenia, AST elevation, creatine phosphokinase elevation, and decreased appetite. The confirmed objective response rate by independent central review was 18.2% (95% CI, 8.2% to 32.7%; five complete responses and three partial responses). The median duration of response was not reached (95% CI, 12.1 weeks to not estimable), and responses were ongoing in six patients (75.0%), including four of five complete responses. Seven of eight responding patients had PD-L1-positive tumors. The median progression-free survival was 11.6 weeks (95% CI, 6.1 to 17.4 weeks); the median OS was 13.7 months (95% CI, 8.5 months to not estimable), with a 12-month OS rate of 54.3% (95% CI, 37.9% to 68.1%). Conclusion Avelumab was well tolerated and associated with durable responses and prolonged survival in patients with refractory metastatic UC.
Figures

References
-
- Ervik M, Lam F, Ferlay J, et al: Cancer Today. Lyon, France: International Agency for Research on Cancer, 2016. http://gco.iarc.fr/today
-
- Kaufman D, Raghavan D, Carducci M, et al. Phase II trial of gemcitabine plus cisplatin in patients with metastatic urothelial cancer. J Clin Oncol. 2000;18:1921–1927. - PubMed
-
- von der Maase H, Hansen SW, Roberts JT, et al. Gemcitabine and cisplatin versus methotrexate, vinblastine, doxorubicin, and cisplatin in advanced or metastatic bladder cancer: Results of a large, randomized, multinational, multicenter, phase III study. J Clin Oncol. 2000;18:3068–3077. - PubMed
-
- Vaughn DJ, Broome CM, Hussain M, et al. Phase II trial of weekly paclitaxel in patients with previously treated advanced urothelial cancer. J Clin Oncol. 2002;20:937–940. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

