A Phase II Study of Dovitinib in Patients with Recurrent or Metastatic Adenoid Cystic Carcinoma
- PMID: 28377480
- PMCID: PMC5540767
- DOI: 10.1158/1078-0432.CCR-16-2942
A Phase II Study of Dovitinib in Patients with Recurrent or Metastatic Adenoid Cystic Carcinoma
Abstract
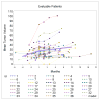
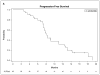
Purpose: Genetic and preclinical studies have implicated FGFR signaling in the pathogenesis of adenoid cystic carcinoma (ACC). Dovitinib, a suppressor of FGFR activity, may be active in ACC.Experimental Design: In a two-stage phase II study, 35 patients with progressive ACC were treated with dovitinib 500 mg orally for 5 of 7 days continuously. The primary endpoints were objective response rate and change in tumor growth rate. Progression-free survival, overall survival, metabolic response, biomarker, and quality of life were secondary endpoints.Results: Of 34 evaluable patients, 2 (6%) had a partial response and 22 (65%) had stable disease >4 months. Median PFS was 8.2 months and OS was 20.6 months. The slope of the overall TGR fell from 1.95 to 0.63 on treatment (P < 0.001). Toxicity was moderate; 63% of patients developed grade 3-4 toxicity, 94% required dose modifications, and 21% stopped treatment early. An early metabolic response based on 18FDG-PET scans was seen in 3 of 15 patients but did not correlate with RECIST response. MYB gene translocation was observed and significantly correlated with overexpression of MYB but did not correlate with FGFR1 phosphorylation or clinical response to dovitinib.Conclusions: Dovitinib produced few objective responses in patients with ACC but did suppress the TGR with a PFS that compares favorably with those reported with other targeted agents. Future studies of more potent and selective FGFR inhibitors in biomarker-selected patients will be required to determine whether FGFR signaling is a valid therapeutic target in ACC. Clin Cancer Res; 23(15); 4138-45. ©2017 AACR.
©2017 American Association for Cancer Research.
Figures


References
-
- Sessions RHL, Forastiere A. Tumors of the salivary glands and paragangliomas. In: Devita VT, Hellman S, Rosenberg SA, editors. Principles and Practice of Oncology. Philadelphia: Lippincott-Raven Publishers; 2000.
-
- Argiris A, Ghebremichael M, Burtness B, Axelrod RS, Deconti RC, Forastiere AA. A phase 2 trial of bortezomib followed by the addition of doxorubicin at progression in patients with recurrent or metastatic adenoid cystic carcinoma of the head and neck: a trial of the Eastern Cooperative Oncology Group (E1303) Cancer. 2011;117(15):3374–3382. - PMC - PubMed
-
- Chau NG, Hotte SJ, Chen EX, Chin SF, Turner S, Wang L, et al. A phase II study of sunitinib in recurrent and/or metastatic adenoid cystic carcinoma (ACC) of the salivary glands: current progress and challenges in evaluating molecularly targeted agents in ACC. Ann Oncol. 2012;23(6):1562–1570. - PubMed
-
- Thomson DJ, Silva P, Denton K, Bonington S, Mak SK, Swindell R, et al. Phase II trial of sorafenib in advanced salivary adenoid cystic carcinoma of the head and neck. Head Neck. 2015;37(2):182–187. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

