Follistatin-like protein 1 modulates IL-17 signaling via IL-17RC regulation in stromal cells
- PMID: 28377613
- PMCID: PMC5609702
- DOI: 10.1038/icb.2017.26
Follistatin-like protein 1 modulates IL-17 signaling via IL-17RC regulation in stromal cells
Abstract
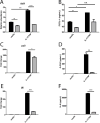
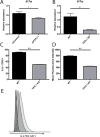
Follistatin-like protein 1 (FSTL-1) possesses several newly identified roles in mammalian biology, including interleukin (IL)-17-driven inflammation, though the mechanism underlying FSTL-1 influence on IL-17-mediated cytokine production is unknown. Using parallel in vitro bone marrow stromal cell models of FSTL-1 suppression, we employed unbiased microarray analysis to identify FSTL-1-regulated genes and pathways that could influence IL-17-dependent production of IL-6 and granulocyte colony-stimulating factor. We discovered that FSTL-1 modulates Il17rc gene expression. Specifically, FSTL-1 was necessary for Il17rc gene transcription, IL-17RC surface protein expression and IL-17-dependent cytokine production. This work identifies a mechanism by which FSTL-1 influences IL-17-driven inflammatory signaling in vitro and reveals a novel function for FSTL-1, as a modulator of gene expression. Thus enhanced understanding of the interplay between FSTL-1 and IL-17-mediated inflammation may provide insight into potential therapeutic targets of IL-17-mediated diseases and warrants ongoing study of in vivo models and clinical scenarios of FSTL-1-influenced diseases.
Figures


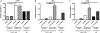

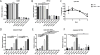
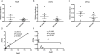
Similar articles
-
FSTL-1 Attenuation Causes Spontaneous Smoke-Resistant Pulmonary Emphysema.Am J Respir Crit Care Med. 2020 Apr 15;201(8):934-945. doi: 10.1164/rccm.201905-0973OC. Am J Respir Crit Care Med. 2020. PMID: 31834999 Free PMC article.
-
Regulation of Pulmonary Bacterial Immunity by Follistatin-Like Protein 1.Infect Immun. 2020 Dec 15;89(1):e00298-20. doi: 10.1128/IAI.00298-20. Print 2020 Dec 15. Infect Immun. 2020. PMID: 33077624 Free PMC article.
-
Follistatin-like protein 1 promotes arthritis by up-regulating IFN-gamma.J Immunol. 2009 Jan 1;182(1):234-9. doi: 10.4049/jimmunol.182.1.234. J Immunol. 2009. PMID: 19109154 Free PMC article.
-
Effects of Moderate Intensity Aerobic Exercise to FSTL-1 Regulation in Atherosclerosis: A Systematic Review.Int J Angiol. 2022 Jul 19;32(1):1-10. doi: 10.1055/s-0042-1750184. eCollection 2023 Mar. Int J Angiol. 2022. PMID: 36727145 Free PMC article. Review.
-
The metabolism-modulating activity of IL-17 signaling in health and disease.J Exp Med. 2021 May 3;218(5):e20202191. doi: 10.1084/jem.20202191. J Exp Med. 2021. PMID: 33822846 Free PMC article. Review.
Cited by
-
Comprehensive profiling of extracellular vesicles in uveitis and scleritis enables biomarker discovery and mechanism exploration.J Transl Med. 2023 Jun 15;21(1):388. doi: 10.1186/s12967-023-04228-x. J Transl Med. 2023. PMID: 37322475 Free PMC article.
-
Potential role of the IL17RC gene in the thoracic ossification of the posterior longitudinal ligament.Int J Mol Med. 2019 May;43(5):2005-2014. doi: 10.3892/ijmm.2019.4130. Epub 2019 Mar 12. Int J Mol Med. 2019. PMID: 30864693 Free PMC article.
-
FSTL-1 Attenuation Causes Spontaneous Smoke-Resistant Pulmonary Emphysema.Am J Respir Crit Care Med. 2020 Apr 15;201(8):934-945. doi: 10.1164/rccm.201905-0973OC. Am J Respir Crit Care Med. 2020. PMID: 31834999 Free PMC article.
-
Regulation of Pulmonary Bacterial Immunity by Follistatin-Like Protein 1.Infect Immun. 2020 Dec 15;89(1):e00298-20. doi: 10.1128/IAI.00298-20. Print 2020 Dec 15. Infect Immun. 2020. PMID: 33077624 Free PMC article.
-
Hepatic expression profiling identifies steatosis-independent and steatosis-driven advanced fibrosis genes.JCI Insight. 2018 Jul 26;3(14):e120274. doi: 10.1172/jci.insight.120274. eCollection 2018 Jul 26. JCI Insight. 2018. PMID: 30046009 Free PMC article.
References
-
- Shibanuma M, Mashimo J, Mita A, Kuroki T, Nose K. Cloning from a mouse osteoblastic cell line of a set of transforming-growth-factor-beta 1-regulated genes, one of which seems to encode a follistatin-related polypeptide. Eur J Biochem. 1993;217(1):13–9. - PubMed
-
- Sylva M, Moorman AF, van den Hoff MJ. Follistatin-like 1 in vertebrate development. Birth defects research Part C, Embryo today : reviews. 2013;99(1):61–9. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous

