Restoring neuronal progranulin reverses deficits in a mouse model of frontotemporal dementia
- PMID: 28379303
- PMCID: PMC5965303
- DOI: 10.1093/brain/awx060
Restoring neuronal progranulin reverses deficits in a mouse model of frontotemporal dementia
Abstract
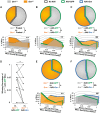
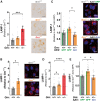
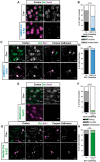
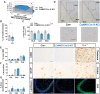
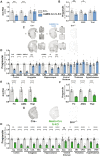
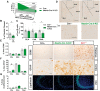
Loss-of-function mutations in progranulin (GRN), a secreted glycoprotein expressed by neurons and microglia, are a common autosomal dominant cause of frontotemporal dementia, a neurodegenerative disease commonly characterized by disrupted social and emotional behaviour. GRN mutations are thought to cause frontotemporal dementia through progranulin haploinsufficiency, therefore, boosting progranulin expression from the intact allele is a rational treatment strategy. However, this approach has not been tested in an animal model of frontotemporal dementia and it is unclear if boosting progranulin could correct pre-existing deficits. Here, we show that adeno-associated virus-driven expression of progranulin in the medial prefrontal cortex reverses social dominance deficits in Grn+/- mice, an animal model of frontotemporal dementia due to GRN mutations. Adeno-associated virus-progranulin also corrected lysosomal abnormalities in Grn+/- mice. The adeno-associated virus-progranulin vector only transduced neurons, suggesting that restoring neuronal progranulin is sufficient to correct deficits in Grn+/- mice. To further test the role of neuronal progranulin in the development of frontotemporal dementia-related deficits, we generated two neuronal progranulin-deficient mouse lines using CaMKII-Cre and Nestin-Cre. Measuring progranulin levels in these lines indicated that most brain progranulin is derived from neurons. Both neuronal progranulin-deficient lines developed social dominance deficits similar to those in global Grn+/- mice, showing that neuronal progranulin deficiency is sufficient to disrupt social behaviour. These data support the concept of progranulin-boosting therapies for frontotemporal dementia and highlight an important role for neuron-derived progranulin in maintaining normal social function.
Keywords: FTD; dementia; experimental models; gene therapy; progranulin.
© The Author (2017). Published by Oxford University Press on behalf of the Guarantors of Brain. All rights reserved. For Permissions, please email: journals.permissions@oup.com.
Figures


References
-
- Baker M, Mackenzie IR, Pickering-Brown SM, Gass J, Rademakers R, Lindholm C. et al. Mutations in progranulin cause tau-negative FTD linked to chromosome 17. Nature 2006; 442: 916–9. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous

