1810011o10 Rik Inhibits the Antitumor Effect of Intratumoral CD8+ T Cells through Suppression of Notch2 Pathway in a Murine Hepatocellular Carcinoma Model
- PMID: 28382040
- PMCID: PMC5360711
- DOI: 10.3389/fimmu.2017.00320
1810011o10 Rik Inhibits the Antitumor Effect of Intratumoral CD8+ T Cells through Suppression of Notch2 Pathway in a Murine Hepatocellular Carcinoma Model
Abstract
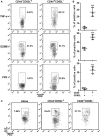
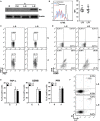
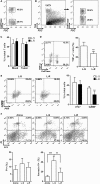
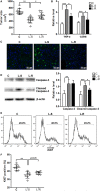
The mechanisms by which tumor-responsive CD8+ T cells are regulated are important for understanding the tumor immunity and for developing new therapeutic strategies. In current study, we identified the expression of 1810011o10 Rik, which is the homolog of human thyroid cancer 1, in intratumoral activated CD8+ T cells in a murine hepatocellular carcinoma (HCC) implantation model. To investigate the role of 1810011o10 Rik in the regulation of antitumor activity of CD8+ T cells, normal CD8+ T cells were transduced with 1810011o10 Rik-expressing lentiviruses. Although 1810011o10 Rik overexpression did not influence agonistic antibody-induced CD8+ T cell activation in vitro, it inhibited the cytotoxic efficacy of CD8+ T cells on HCC cells in vivo. 1810011o10 Rik overexpression impeded CD8+ T cell-mediated HCC cell apoptosis and favored tumor cell growth in vivo. Further investigation revealed that 1810011o10 Rik blocked the nuclear translocation of Notch2 intracellular domain, which is crucial for CD8+ T cell activity. Furthermore, a brief in vitro experiment suggested that both antigen-presenting cells and TGF-β might be necessary for the upregulation of Rik expression in activated CD8+ T cells. In general, our study disclosed a novel mechanism underlying the negative regulation of antitumor CD8+ T cells during HCC progression.
Keywords: 1810011o10 Rik; CD8+ T cells; Notch2; hepatocellular carcinoma; immunity.
Figures



Similar articles
-
Amphiregulin activates regulatory T lymphocytes and suppresses CD8+ T cell-mediated anti-tumor response in hepatocellular carcinoma cells.Oncotarget. 2015 Oct 13;6(31):32138-53. doi: 10.18632/oncotarget.5171. Oncotarget. 2015. PMID: 26451607 Free PMC article.
-
NKT and CD8 lymphocytes mediate suppression of hepatocellular carcinoma growth via tumor antigen-pulsed dendritic cells.Int J Cancer. 2003 Aug 20;106(2):236-43. doi: 10.1002/ijc.11201. Int J Cancer. 2003. PMID: 12800200
-
Interferon-gamma produced by interleukin-12-activated tumor infiltrating CD8+T cells directly induces apoptosis of mouse hepatocellular carcinoma.J Hepatol. 2006 Nov;45(5):662-72. doi: 10.1016/j.jhep.2006.05.018. Epub 2006 Jul 7. J Hepatol. 2006. PMID: 16935390
-
Specific CD8(+) T cell response immunotherapy for hepatocellular carcinoma and viral hepatitis.World J Gastroenterol. 2016 Jul 28;22(28):6469-83. doi: 10.3748/wjg.v22.i28.6469. World J Gastroenterol. 2016. PMID: 27605882 Free PMC article. Review.
-
Pleiotropic effects of methionine adenosyltransferases deregulation as determinants of liver cancer progression and prognosis.J Hepatol. 2013 Oct;59(4):830-41. doi: 10.1016/j.jhep.2013.04.031. Epub 2013 May 7. J Hepatol. 2013. PMID: 23665184 Review.
Cited by
-
Whole Exome Sequencing of Intracranial Epidermoid Cysts Reveals Immune-Associated Mechanistic and Potential Targets.Cancers (Basel). 2024 Oct 15;16(20):3487. doi: 10.3390/cancers16203487. Cancers (Basel). 2024. PMID: 39456581 Free PMC article.
-
Notch1 blockade by a novel, selective anti-Notch1 neutralizing antibody improves immunotherapy efficacy in melanoma by promoting an inflamed TME.J Exp Clin Cancer Res. 2024 Nov 4;43(1):295. doi: 10.1186/s13046-024-03214-5. J Exp Clin Cancer Res. 2024. PMID: 39491031 Free PMC article.
-
MS-20 enhances the gut microbiota-associated antitumor effects of anti-PD1 antibody.Gut Microbes. 2024 Jan-Dec;16(1):2380061. doi: 10.1080/19490976.2024.2380061. Epub 2024 Jul 30. Gut Microbes. 2024. PMID: 39078050 Free PMC article.
-
The role of oncogenic Notch2 signaling in cancer: a novel therapeutic target.Am J Cancer Res. 2019 May 1;9(5):837-854. eCollection 2019. Am J Cancer Res. 2019. PMID: 31218097 Free PMC article. Review.
-
Gliotoxin Targets Nuclear NOTCH2 in Human Solid Tumor Derived Cell Lines In Vitro and Inhibits Melanoma Growth in Xenograft Mouse Model.Front Pharmacol. 2017 Jul 7;8:319. doi: 10.3389/fphar.2017.00319. eCollection 2017. Front Pharmacol. 2017. PMID: 28736522 Free PMC article.
References
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

