Concurrent tACS-fMRI Reveals Causal Influence of Power Synchronized Neural Activity on Resting State fMRI Connectivity
- PMID: 28385876
- PMCID: PMC6596494
- DOI: 10.1523/JNEUROSCI.1756-16.2017
Concurrent tACS-fMRI Reveals Causal Influence of Power Synchronized Neural Activity on Resting State fMRI Connectivity
Abstract
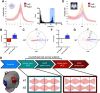
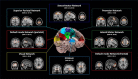
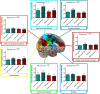
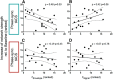
Resting state fMRI (rs-fMRI) is commonly used to study the brain's intrinsic neural coupling, which reveals specific spatiotemporal patterns in the form of resting state networks (RSNs). It has been hypothesized that slow rs-fMRI oscillations (<0.1 Hz) are driven by underlying electrophysiological rhythms that typically occur at much faster timescales (>5 Hz); however, causal evidence for this relationship is currently lacking. Here we measured rs-fMRI in humans while applying transcranial alternating current stimulation (tACS) to entrain brain rhythms in left and right sensorimotor cortices. The two driving tACS signals were tailored to the individual's α rhythm (8-12 Hz) and fluctuated in amplitude according to a 1 Hz power envelope. We entrained the left versus right hemisphere in accordance to two different coupling modes where either α oscillations were synchronized between hemispheres (phase-synchronized tACS) or the slower oscillating power envelopes (power-synchronized tACS). Power-synchronized tACS significantly increased rs-fMRI connectivity within the stimulated RSN compared with phase-synchronized or no tACS. This effect outlasted the stimulation period and tended to be more effective in individuals who exhibited a naturally weak interhemispheric coupling. Using this novel approach, our data provide causal evidence that synchronized power fluctuations contribute to the formation of fMRI-based RSNs. Moreover, our findings demonstrate that the brain's intrinsic coupling at rest can be selectively modulated by choosing appropriate tACS signals, which could lead to new interventions for patients with altered rs-fMRI connectivity.SIGNIFICANCE STATEMENT Resting state fMRI (rs-fMRI) has become an important tool to estimate brain connectivity. However, relatively little is known about how slow hemodynamic oscillations measured with fMRI relate to electrophysiological processes. It was suggested that slowly fluctuating power envelopes of electrophysiological signals synchronize across brain areas and that the topography of this activity is spatially correlated to resting state networks derived from rs-fMRI. Here we take a novel approach to address this problem and establish a causal link between the power fluctuations of electrophysiological signals and rs-fMRI via a new neuromodulation paradigm, which exploits these power synchronization mechanisms. These novel mechanistic insights bridge different scientific domains and are of broad interest to researchers in the fields of Medical Imaging, Neuroscience, Physiology, and Psychology.
Keywords: EEG; electrical stimulation; mechanism; neuronal oscillations; simultaneous tACS/fMRI.
Copyright © 2017 the authors 0270-6474/17/374766-12$15.00/0.
Figures





References
-
- Beckmann CF, Mackay CE, Filippini N, Smith SM (2009) Group comparison of resting-state fMRI data using multi-subject ICA and dual regression. Neuroimage 47:S148 10.1016/S1053-8119(09)71514-9 - DOI
-
- Berens P. (2009) CircStat: a MATLABToolbox for circular statistics. J Statist Software 31:1–21.
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
