Bilayer-Mediated Structural Transitions Control Mechanosensitivity of the TREK-2 K2P Channel
- PMID: 28392258
- PMCID: PMC5415359
- DOI: 10.1016/j.str.2017.03.006
Bilayer-Mediated Structural Transitions Control Mechanosensitivity of the TREK-2 K2P Channel
Abstract
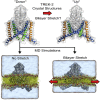
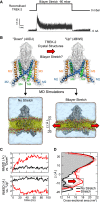
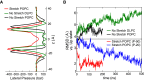
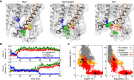
The mechanosensitive two-pore domain (K2P) K+ channels (TREK-1, TREK-2, and TRAAK) are important for mechanical and thermal nociception. However, the mechanisms underlying their gating by membrane stretch remain controversial. Here we use molecular dynamics simulations to examine their behavior in a lipid bilayer. We show that TREK-2 moves from the "down" to "up" conformation in direct response to membrane stretch, and examine the role of the transmembrane pressure profile in this process. Furthermore, we show how state-dependent interactions with lipids affect the movement of TREK-2, and how stretch influences both the inner pore and selectivity filter. Finally, we present functional studies that demonstrate why direct pore block by lipid tails does not represent the principal mechanism of mechanogating. Overall, this study provides a dynamic structural insight into K2P channel mechanosensitivity and illustrates how the structure of a eukaryotic mechanosensitive ion channel responds to changes in forces within the bilayer.
Keywords: K(+) channel gating; K2P channel; KCNK10; KCNK2; KCNK4; Mechanosensitive; TREK-2.
Copyright © 2017 The Authors. Published by Elsevier Ltd.. All rights reserved.
Figures




Similar articles
-
Negative Influence by the Force: Mechanically Induced Hyperpolarization via K2P Background Potassium Channels.Int J Mol Sci. 2021 Aug 23;22(16):9062. doi: 10.3390/ijms22169062. Int J Mol Sci. 2021. PMID: 34445768 Free PMC article. Review.
-
Asymmetric mechanosensitivity in a eukaryotic ion channel.Proc Natl Acad Sci U S A. 2017 Oct 3;114(40):E8343-E8351. doi: 10.1073/pnas.1708990114. Epub 2017 Sep 18. Proc Natl Acad Sci U S A. 2017. PMID: 28923939 Free PMC article.
-
The effects of stretch activation on ionic selectivity of the TREK-2 K2P K+ channel.Channels (Austin). 2017 Sep 3;11(5):482-486. doi: 10.1080/19336950.2017.1356955. Epub 2017 Jul 19. Channels (Austin). 2017. PMID: 28723241 Free PMC article.
-
Stretch-activated two-pore-domain (K2P) potassium channels in the heart: Focus on atrial fibrillation and heart failure.Prog Biophys Mol Biol. 2017 Nov;130(Pt B):233-243. doi: 10.1016/j.pbiomolbio.2017.05.004. Epub 2017 May 16. Prog Biophys Mol Biol. 2017. PMID: 28526353
-
Temperature sensitivity of two-pore (K2P) potassium channels.Curr Top Membr. 2014;74:113-33. doi: 10.1016/B978-0-12-800181-3.00005-1. Curr Top Membr. 2014. PMID: 25366235 Free PMC article. Review.
Cited by
-
Structural Insights into the Mechanisms and Pharmacology of K2P Potassium Channels.J Mol Biol. 2021 Aug 20;433(17):166995. doi: 10.1016/j.jmb.2021.166995. Epub 2021 Apr 20. J Mol Biol. 2021. PMID: 33887333 Free PMC article. Review.
-
From Bench to Biomolecular Simulation: Phospholipid Modulation of Potassium Channels.J Mol Biol. 2021 Aug 20;433(17):167105. doi: 10.1016/j.jmb.2021.167105. Epub 2021 Jun 15. J Mol Biol. 2021. PMID: 34139216 Free PMC article. Review.
-
Atomistic mechanism of coupling between cytosolic sensor domain and selectivity filter in TREK K2P channels.Nat Commun. 2024 May 31;15(1):4628. doi: 10.1038/s41467-024-48823-y. Nat Commun. 2024. PMID: 38821927 Free PMC article.
-
Protein-lipid charge interactions control the folding of outer membrane proteins into asymmetric membranes.Nat Chem. 2023 Dec;15(12):1754-1764. doi: 10.1038/s41557-023-01319-6. Epub 2023 Sep 14. Nat Chem. 2023. PMID: 37710048 Free PMC article.
-
Negative Influence by the Force: Mechanically Induced Hyperpolarization via K2P Background Potassium Channels.Int J Mol Sci. 2021 Aug 23;22(16):9062. doi: 10.3390/ijms22169062. Int J Mol Sci. 2021. PMID: 34445768 Free PMC article. Review.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

