A mutation in the Tubb4a gene leads to microtubule accumulation with hypomyelination and demyelination
- PMID: 28393430
- PMCID: PMC5495199
- DOI: 10.1002/ana.24930
A mutation in the Tubb4a gene leads to microtubule accumulation with hypomyelination and demyelination
Abstract
Objective: Our goal was to define the genetic cause of the profound hypomyelination in the taiep rat model and determine its relevance to human white matter disease.
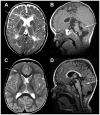
Methods: Based on previous localization of the taiep mutation to rat chromosome 9, we tested whether the mutation resided within the Tubb4a (β-tubulin 4A) gene, because mutations in the TUBB4A gene have been described in patients with central nervous system hypomyelination. To determine whether accumulation of microtubules led to progressive demyelination, we analyzed the spinal cord and optic nerves of 2-year-old rats by light and electron microscopy. Cerebral white matter from a patient with TUBB4A Asn414Lys mutation and magnetic resonance imaging evidence of severe hypomyelination were studied similarly.
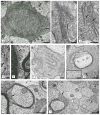
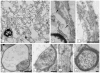
Results: As the taiep rat ages, there is progressive loss of myelin in the brain and dorsal column of the spinal cord associated with increased oligodendrocyte numbers with accumulation of microtubules. This accumulation involved the entire cell body and distal processes of oligodendrocytes, but there was no accumulation of microtubules in axons. A single point mutation in Tubb4a (p.Ala302Thr) was found in homozygous taiep samples. A similar hypomyelination associated with increased oligodendrocyte numbers and arrays of microtubules in oligodendrocytes was demonstrated in the human patient sample.
Interpretation: The taiep rat is the first animal model of TUBB4 mutations in humans and a novel system in which to test the mechanism of microtubule accumulation. The finding of microtubule accumulation in a patient with a TUBB4A mutation and leukodystrophy confirms the usefulness of taiep as a model of the human disease. Ann Neurol 2017;81:690-702.
© 2017 American Neurological Association.
Conflict of interest statement
None of the authors have any conflict of interest.
Figures
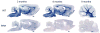




Similar articles
-
Microtubule alterations in cultured taiep rat oligodendrocytes lead to deficits in myelin membrane formation.J Neurocytol. 1999 Aug;28(8):671-83. doi: 10.1023/a:1007060832459. J Neurocytol. 1999. PMID: 10851346
-
TUBB4A mutations result in specific neuronal and oligodendrocytic defects that closely match clinically distinct phenotypes.Hum Mol Genet. 2017 Nov 15;26(22):4506-4518. doi: 10.1093/hmg/ddx338. Hum Mol Genet. 2017. PMID: 28973395 Free PMC article.
-
Dysmyelination, demyelination and reactive astrogliosis in the optic nerve of the taiep rat.Biol Res. 1999;32(4):253-62. doi: 10.4067/s0716-97601999000400005. Biol Res. 1999. PMID: 10983245
-
[A report of atypical hypomyelinating leukodystrophy with atrophy of the basal ganglia and cerebellum caused by a de novo mutation in tubulin beta 4A (TUBB4A) gene and literature review].Zhonghua Nei Ke Za Zhi. 2017 Jun 1;56(6):433-437. doi: 10.3760/cma.j.issn.0578-1426.2017.06.009. Zhonghua Nei Ke Za Zhi. 2017. PMID: 28592043 Review. Chinese.
-
Novel TUBB4A mutations and expansion of the neuroimaging phenotype of hypomyelination with atrophy of the basal ganglia and cerebellum (H-ABC).Am J Med Genet A. 2014 Jul;164A(7):1802-7. doi: 10.1002/ajmg.a.36526. Epub 2014 Apr 4. Am J Med Genet A. 2014. PMID: 24706558 Free PMC article. Review.
Cited by
-
Characterization of sperm motility and testosterone secretion in the taiep myelin mutant, a model of demyelination.Anim Reprod. 2023 Oct 30;20(3):e20220102. doi: 10.1590/1984-3143-AR2022-0102. eCollection 2023. Anim Reprod. 2023. PMID: 38026000 Free PMC article.
-
Hypomyelinating leukodystrophies - unravelling myelin biology.Nat Rev Neurol. 2021 Feb;17(2):88-103. doi: 10.1038/s41582-020-00432-1. Epub 2020 Dec 15. Nat Rev Neurol. 2021. PMID: 33324001 Review.
-
The Golgi Outpost Protein TPPP Nucleates Microtubules and Is Critical for Myelination.Cell. 2019 Sep 19;179(1):132-146.e14. doi: 10.1016/j.cell.2019.08.025. Epub 2019 Sep 12. Cell. 2019. PMID: 31522887 Free PMC article.
-
Functional Investigation of TUBB4A Variants Associated with Different Clinical Phenotypes.Mol Neurobiol. 2022 Aug;59(8):5056-5069. doi: 10.1007/s12035-022-02900-9. Epub 2022 Jun 6. Mol Neurobiol. 2022. PMID: 35668344
-
Multi-Omics Analysis of Hippocampus in Rats Administered Trimethyltin Chloride.Neurotox Res. 2025 Mar 17;43(2):13. doi: 10.1007/s12640-025-00737-3. Neurotox Res. 2025. PMID: 40095120 Free PMC article.
References
-
- van der Knaap MS, Linnankivi T, Paetau A, et al. Hypomyelination with atrophy of the basal ganglia and cerebellum: follow-up and pathology. Neurology. 2007 Jul 10;69(2):166–71. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials

