Engineering plant membranes using droplet interface bilayers
- PMID: 28396711
- PMCID: PMC5367087
- DOI: 10.1063/1.4979045
Engineering plant membranes using droplet interface bilayers
Abstract
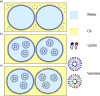
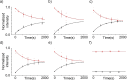
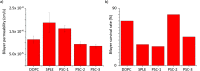
Droplet interface bilayers (DIBs) have become widely recognised as a robust platform for constructing model membranes and are emerging as a key technology for the bottom-up assembly of synthetic cell-like and tissue-like structures. DIBs are formed when lipid-monolayer coated water droplets are brought together inside a well of oil, which is excluded from the interface as the DIB forms. The unique features of the system, compared to traditional approaches (e.g., supported lipid bilayers, black lipid membranes, and liposomes), is the ability to engineer multi-layered bilayer networks by connecting multiple droplets together in 3D, and the capability to impart bilayer asymmetry freely within these droplet architectures by supplying droplets with different lipids. Yet despite these achievements, one potential limitation of the technology is that DIBs formed from biologically relevant components have not been well studied. This could limit the reach of the platform to biological systems where bilayer composition and asymmetry are understood to play a key role. Herein, we address this issue by reporting the assembly of asymmetric DIBs designed to replicate the plasma membrane compositions of three different plant species; Arabidopsis thaliana, tobacco, and oats, by engineering vesicles with different amounts of plant phospholipids, sterols and cerebrosides for the first time. We show that vesicles made from our plant lipid formulations are stable and can be used to assemble asymmetric plant DIBs. We verify this using a bilayer permeation assay, from which we extract values for absolute effective bilayer permeation and bilayer stability. Our results confirm that stable DIBs can be assembled from our plant membrane mimics and could lead to new approaches for assembling model systems to study membrane translocation and to screen new agrochemicals in plants.
Figures


Similar articles
-
Temperature-Controlled Assembly and Characterization of a Droplet Interface Bilayer.J Vis Exp. 2021 Apr 19;(170). doi: 10.3791/62362. J Vis Exp. 2021. PMID: 33938884
-
Heating-enabled formation of droplet interface bilayers using Escherichia coli total lipid extract.Langmuir. 2015;31(1):325-37. doi: 10.1021/la503471m. Epub 2014 Dec 31. Langmuir. 2015. PMID: 25514167
-
Adsorption Kinetics Dictate Monolayer Self-Assembly for Both Lipid-In and Lipid-Out Approaches to Droplet Interface Bilayer Formation.Langmuir. 2015 Dec 1;31(47):12883-93. doi: 10.1021/acs.langmuir.5b02293. Epub 2015 Nov 19. Langmuir. 2015. PMID: 26556227
-
Physicochemical characteristics of droplet interface bilayers.Adv Colloid Interface Sci. 2022 Jun;304:102666. doi: 10.1016/j.cis.2022.102666. Epub 2022 Apr 8. Adv Colloid Interface Sci. 2022. PMID: 35429720 Review.
-
Lipid Bilayers Manipulated through Monolayer Technologies for Studies of Channel-Membrane Interplay.Biol Pharm Bull. 2018;41(3):303-311. doi: 10.1248/bpb.b17-00708. Biol Pharm Bull. 2018. PMID: 29491206 Review.
Cited by
-
Membrane protein mediated bilayer communication in networks of droplet interface bilayers.Commun Chem. 2020 Jun 12;3:77. doi: 10.1038/s42004-020-0322-1. eCollection 2020 Jun 12. Commun Chem. 2020. PMID: 34113722 Free PMC article.
-
Probing membrane protein properties using droplet interface bilayers.Exp Biol Med (Maywood). 2019 May;244(8):709-720. doi: 10.1177/1535370219847939. Epub 2019 May 3. Exp Biol Med (Maywood). 2019. PMID: 31053046 Free PMC article.
-
Challenges and opportunities in achieving the full potential of droplet interface bilayers.Nat Chem. 2022 Aug;14(8):862-870. doi: 10.1038/s41557-022-00989-y. Epub 2022 Jul 25. Nat Chem. 2022. PMID: 35879442 Review.
References
LinkOut - more resources
Full Text Sources
Other Literature Sources
