A Novel Strategy to Co-target Estrogen Receptor and Nuclear Factor κB Pathways with Hybrid Drugs for Breast Cancer Therapy
- PMID: 28396978
- PMCID: PMC5690576
- DOI: 10.1007/s12672-017-0294-5
A Novel Strategy to Co-target Estrogen Receptor and Nuclear Factor κB Pathways with Hybrid Drugs for Breast Cancer Therapy
Abstract
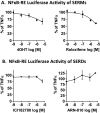
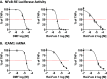
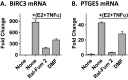
Nearly 75% of breast tumors express estrogen receptor (ER), and will be treated with endocrine therapy, such as selective estrogen receptor modulator (SERM), tamoxifen, or aromatase inhibitors. Despite their proven success, as many as 40-50% of ER+ tumors fail to respond to endocrine therapy and eventually recur as aggressive, metastatic cancers. Therefore, preventing and/or overcoming endocrine resistance in ER+ tumors remains a major clinical challenge. Deregulation or activation of the nuclear factor κB (NFκB) pathway has been implicated in endocrine resistance and poor patient outcome in ER+ tumors. As a consequence, one option to improve on existing anti-cancer treatment regimens may be to introduce additional anti-NFκB activity to endocrine therapy drugs. Our approach was to design and test SERM-fumarate co-targeting hybrid drugs capable of simultaneously inhibiting both ER, via the SERM, raloxifene, and the NFκB pathway, via fumarate, in breast cancer cells. We find that the hybrid drugs display improved anti-NFκB pathway inhibition compared to either raloxifene or fumarate. Despite some loss in potency against the ER pathway, these hybrid drugs maintain anti-proliferative activity in ER+ breast cancer cells. Furthermore, these drugs prevent clonogenic growth and mammosphere formation of ER+ breast cancer cells. As a proof-of-principle, the simultaneous inhibition of ER and NFκB via a single bifunctional hybrid drug may represent a viable approach to improve the anti-inflammatory activity and prevent therapy resistance of ER-targeted anti-cancer drugs.
Keywords: Breast cancer; Co-targeting drugs; Estrogen receptor; NFκB pathway.
Conflict of interest statement
The authors declare that they have no conflict of interest.
Figures

Similar articles
-
The NFkappaB pathway and endocrine-resistant breast cancer.Endocr Relat Cancer. 2005 Jul;12 Suppl 1:S37-46. doi: 10.1677/erc.1.00977. Endocr Relat Cancer. 2005. PMID: 16113098 Review.
-
Update on the Role of NFκB in Promoting Aggressive Phenotypes of Estrogen Receptor-Positive Breast Cancer.Endocrinology. 2020 Oct 1;161(10):bqaa152. doi: 10.1210/endocr/bqaa152. Endocrinology. 2020. PMID: 32887995 Free PMC article. Review.
-
Therapeutic targeting in the estrogen receptor hormonal pathway.Semin Oncol. 2004 Feb;31(1 Suppl 3):28-38. doi: 10.1053/j.seminoncol.2004.01.004. Semin Oncol. 2004. PMID: 15052541 Review.
-
Selective pressure of endocrine therapy activates the integrated stress response through NFκB signaling in a subpopulation of ER positive breast cancer cells.Breast Cancer Res. 2022 Mar 9;24(1):19. doi: 10.1186/s13058-022-01515-1. Breast Cancer Res. 2022. PMID: 35264224 Free PMC article.
-
A novel aspirin prodrug inhibits NFκB activity and breast cancer stem cell properties.BMC Cancer. 2015 Nov 4;15:845. doi: 10.1186/s12885-015-1868-7. BMC Cancer. 2015. PMID: 26530254 Free PMC article.
Cited by
-
Pharmacological, Mechanistic, and Pharmacokinetic Assessment of Novel Melatonin-Tamoxifen Drug Conjugates as Breast Cancer Drugs.Mol Pharmacol. 2019 Aug;96(2):272-296. doi: 10.1124/mol.119.116202. Epub 2019 Jun 20. Mol Pharmacol. 2019. PMID: 31221824 Free PMC article.
-
Dexamethasone and Fumaric Acid Ester Conjugate Synergistically Inhibits Inflammation and NF-κB in Macrophages.Bioconjug Chem. 2021 Aug 18;32(8):1629-1640. doi: 10.1021/acs.bioconjchem.1c00200. Epub 2021 Jun 24. Bioconjug Chem. 2021. PMID: 34165285 Free PMC article.
-
Drug conjugates-an emerging approach to treat breast cancer.Pharmacol Res Perspect. 2018 Jul 5;6(4):e00417. doi: 10.1002/prp2.417. eCollection 2018 Jul. Pharmacol Res Perspect. 2018. PMID: 29983986 Free PMC article. Review.
-
Five-year survival in luminal breast cancer patients: relation with intratumoral activity of proteasomes.Transl Breast Cancer Res. 2022 Jul 30;3:23. doi: 10.21037/tbcr-22-22. eCollection 2022. Transl Breast Cancer Res. 2022. PMID: 38751528 Free PMC article.
References
-
- Jones RL, Rojo F, A'Hern R, Villena N, Salter J, Corominas JM, Servitja S, Smith IE, Rovira A, Reis-Filho JS, Dowsett M, Albanell J. Nuclear NF-kappaB/p65 expression and response to neoadjuvant chemotherapy in breast cancer. J Clin Pathol. 2011;64:130–135. doi: 10.1136/jcp.2010.082966. - DOI - PubMed
-
- Kubo M, Kanaya N, Petrossian K, Ye J, Warden C, Liu Z, Nishimura R, Osako T, Okido M, Shimada K, Takahashi M, Chu P, Yuan YC, Chen S. Inhibition of the proliferation of acquired aromatase inhibitor-resistant breast cancer cells by histone deacetylase inhibitor LBH589 (panobinostat) Breast Cancer Res Treat. 2013;137:93–107. doi: 10.1007/s10549-012-2332-x. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

