Nanoparticulate Tetrac Inhibits Growth and Vascularity of Glioblastoma Xenografts
- PMID: 28396979
- PMCID: PMC5413536
- DOI: 10.1007/s12672-017-0293-6
Nanoparticulate Tetrac Inhibits Growth and Vascularity of Glioblastoma Xenografts
Abstract
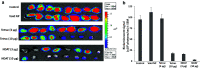
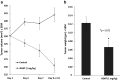
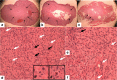
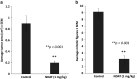
Thyroid hormone as L-thyroxine (T4) stimulates proliferation of glioma cells in vitro and medical induction of hypothyroidism slows clinical growth of glioblastoma multiforme (GBM). The proliferative action of T4 on glioma cells is initiated nongenomically at a cell surface receptor for thyroid hormone on the extracellular domain of integrin αvβ3. Tetraiodothyroacetic acid (tetrac) is a thyroid hormone derivative that blocks T4 action at αvβ3 and has anticancer and anti-angiogenic activity. Tetrac has been covalently bonded via a linker to a nanoparticle (Nanotetrac, Nano-diamino-tetrac, NDAT) that increases the potency of tetrac and broadens the anticancer properties of the drug. In the present studies of human GBM xenografts in immunodeficient mice, NDAT administered daily for 10 days subcutaneously as 1 mg tetrac equivalent/kg reduced tumor xenograft weight at animal sacrifice by 50%, compared to untreated control lesions (p < 0.01). Histopathological analysis of tumors revealed a 95% loss of the vascularity of treated tumors compared to controls at 10 days (p < 0.001), without intratumoral hemorrhage. Up to 80% of tumor cells were necrotic in various microscopic fields (p < 0.001 vs. control tumors), an effect attributable to devascularization. There was substantial evidence of apoptosis in other fields (p < 0.001 vs. control tumors). Induction of apoptosis in cancer cells is a well-described quality of NDAT. In summary, systemic NDAT has been shown to be effective by multiple mechanisms in treatment of GBM xenografts.
Conflict of interest statement
Co-authors Davis and Mousa are stockholders in NanoPharmaceuticals LLC that is commercially developing NDAT (Nanotetrac, Nano-diamino-tetrac) and Davis is an officer of the company. All remaining authors have no conflicts to declare.
Figures


Similar articles
-
Tetraidothyroacetic acid (tetrac) and tetrac nanoparticles inhibit growth of human renal cell carcinoma xenografts.Anticancer Res. 2009 Oct;29(10):3825-31. Anticancer Res. 2009. PMID: 19846915
-
Pharmacokinetics, Biodistribution, and Anti-Angiogenesis Efficacy of Diamino Propane Tetraiodothyroacetic Acid-conjugated Biodegradable Polymeric Nanoparticle.Sci Rep. 2019 Jun 21;9(1):9006. doi: 10.1038/s41598-019-44979-6. Sci Rep. 2019. Retraction in: Sci Rep. 2023 Oct 3;13(1):16594. doi: 10.1038/s41598-023-43642-5. PMID: 31227723 Free PMC article. Retracted.
-
Tetraiodothyroacetic acid and its nanoformulation inhibit thyroid hormone stimulation of non-small cell lung cancer cells in vitro and its growth in xenografts.Lung Cancer. 2012 Apr;76(1):39-45. doi: 10.1016/j.lungcan.2011.10.003. Epub 2011 Oct 22. Lung Cancer. 2012. PMID: 22024450
-
Actions of L-thyroxine (T4) and Tetraiodothyroacetic Acid (Tetrac) on Gene Expression in Thyroid Cancer Cells.Genes (Basel). 2020 Jul 7;11(7):755. doi: 10.3390/genes11070755. Genes (Basel). 2020. PMID: 32645835 Free PMC article. Review.
-
Cancer Cell Gene Expression Modulated from Plasma Membrane Integrin αvβ3 by Thyroid Hormone and Nanoparticulate Tetrac.Front Endocrinol (Lausanne). 2015 Jan 12;5:240. doi: 10.3389/fendo.2014.00240. eCollection 2014. Front Endocrinol (Lausanne). 2015. PMID: 25628605 Free PMC article. Review.
Cited by
-
Evidence of Aquaporin 4 Regulation by Thyroid Hormone During Mouse Brain Development and in Cultured Human Glioblastoma Multiforme Cells.Front Neurosci. 2019 Apr 4;13:317. doi: 10.3389/fnins.2019.00317. eCollection 2019. Front Neurosci. 2019. PMID: 31019448 Free PMC article.
-
Main Factors Involved in Thyroid Hormone Action.Molecules. 2021 Dec 3;26(23):7337. doi: 10.3390/molecules26237337. Molecules. 2021. PMID: 34885918 Free PMC article. Review.
-
Nano-Therapies for Glioblastoma Treatment.Cancers (Basel). 2020 Jan 19;12(1):242. doi: 10.3390/cancers12010242. Cancers (Basel). 2020. PMID: 31963825 Free PMC article. Review.
-
Novel Targeted Nano-Parthenolide Molecule against NF-kB in Acute Myeloid Leukemia.Molecules. 2019 Jun 3;24(11):2103. doi: 10.3390/molecules24112103. Molecules. 2019. PMID: 31163672 Free PMC article.
-
Thyroid Hormones and Cancer: A Comprehensive Review of Preclinical and Clinical Studies.Front Endocrinol (Lausanne). 2019 Feb 13;10:59. doi: 10.3389/fendo.2019.00059. eCollection 2019. Front Endocrinol (Lausanne). 2019. PMID: 30814976 Free PMC article. Review.
References
-
- Mrugala MM. Advances and challenges in the treatment of glioblastoma: a clinician’s perspective. Discov Med. 2013;15(83):221–230. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

