Cholinergic agonists reduce blood pressure in a mouse model of systemic lupus erythematosus
- PMID: 28400502
- PMCID: PMC5392509
- DOI: 10.14814/phy2.13213
Cholinergic agonists reduce blood pressure in a mouse model of systemic lupus erythematosus
Abstract
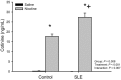
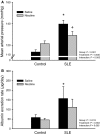
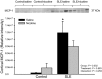
Increased inflammation arising from an abnormal immune response can damage healthy tissue and lead to disease progression. An important example of this is the accumulation of inflammatory mediators in the kidney, which can subsequently lead to hypertension and renal injury. The origin of this inflammation may involve neuro-immune interactions. For example, the novel vagus nerve-to-spleen mechanism known as the "cholinergic anti-inflammatory pathway" controls inflammation upon stimulation. However, if this pathway is dysfunctional, inflammation becomes less regulated and chronic inflammatory diseases such as hypertension may develop. Systemic lupus erythematosus (SLE) is an autoimmune disease with aberrant immune function, increased renal inflammation, and prevalent hypertension. We hypothesized that the cholinergic anti-inflammatory pathway is impaired in SLE and that stimulation of this pathway would protect from the progression of hypertension in SLE mice. Female SLE (NZBWF1) and control (NZW) mice were administered nicotine or vehicle for 7 days (2 mg/kg/day, subcutaneously) in order to stimulate the cholinergic anti-inflammatory pathway at the level of the splenic nicotinic acetylcholine receptor (α7-nAChR). Blood pressure was assessed posttreatment. Nicotine-treated SLE mice did not develop hypertension and this lower blood pressure (compared to saline-treated SLE mice) coincided with lower splenic and renal cortical expression of pro-inflammatory cytokines. These data provide evidence that the cholinergic anti-inflammatory pathway is impaired in SLE In addition, these data suggest that stimulation of the cholinergic anti-inflammatory pathway can protect the kidney by dampening inflammation and therefore prevent the progression of hypertension in the setting of SLE.
Keywords: Cholinergic anti‐inflammatory pathway; hypertension; inflammation; kidney; nicotine; renal injury.
© 2017 The Authors. Physiological Reports published by Wiley Periodicals, Inc. on behalf of The Physiological Society and the American Physiological Society.
Figures



References
-
- Agarwal, P. K. , van den Born J., van Goor H., Navis G., Gans R. O. B., and Bakker S. J. L.. 2012. Renoprotective effects of long‐term oral nicotine in a rat model of spontaneous proteinuria. Am. J. Physiol. Renal. Physiol. 302:895–904. - PubMed
-
- Al‐Herz, A. , Ensworth S., Shojania K., and Esdaile J.. 2003. Cardiovascular risk factor screening in systemic lupus erythematosus. J. Rheumatol. 30:493–496. - PubMed
-
- Borovikova, L. , Ivanova S., Nardi D., Zhang M., Yang H., Ombrellino M., et al. 2000a. Role of vagus nerve signaling in CNI‐1493‐mediated suppression of acute inflammation. Auto. Neurosci. 85:141–147. - PubMed
-
- Borovikova, L. , Ivanova S., Zhang M., Yang H., Botchkina G., Watkins L., et al. 2000b. Vagus nerve stimulation attenuates the systemic inflammatory response to endotoxin. Nature 405:458–462. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

