Role of Modulator of Inflammation Cyclooxygenase-2 in Gammaherpesvirus Mediated Tumorigenesis
- PMID: 28400769
- PMCID: PMC5368278
- DOI: 10.3389/fmicb.2017.00538
Role of Modulator of Inflammation Cyclooxygenase-2 in Gammaherpesvirus Mediated Tumorigenesis
Abstract
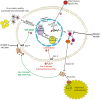
Chronic inflammation is recognized as a threat factor for cancer progression. Release of inflammatory molecules generates microenvironment which is highly favorable for development of tumor, cancer progression and metastasis. In cases of latent viral infections, generation of such a microenvironment is one of the major predisposing factors related to virus mediated tumorigenesis. Among various inflammatory mediators implicated in pathological process associated with cancer, the cyclooxygenase (COX) and its downstream effector molecules are of greater significance. Though the role of infectious agents in causing inflammation leading to transformation of cells has been more or less well established, however, the mechanism by which inflammation in itself modulates the events in life cycle of infectious agent is not very much clear. This is specifically important for gammaherpesviruses infections where viral life cycle is characterized by prolonged periods of latency when the virus remains hidden, immunologically undetectable and expresses only a very limited set of genes. Therefore, it is important to understand the mechanisms for role of inflammation in virus life cycle and tumorigenesis. This review is an attempt to summarize the latest findings highlighting the significance of COX-2 and its downstream signaling effectors role in life cycle events of gammaherpesviruses leading to progression of cancer.
Keywords: COX-2; gammaherpesvirus; inflammation.
Figures

References
Publication types
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

