Constitutive activation of kappa opioid receptors at ventral tegmental area inhibitory synapses following acute stress
- PMID: 28402252
- PMCID: PMC5389861
- DOI: 10.7554/eLife.23785
Constitutive activation of kappa opioid receptors at ventral tegmental area inhibitory synapses following acute stress
Abstract
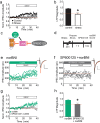
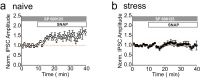
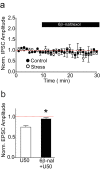
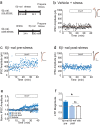
Stressful experiences potently activate kappa opioid receptors (κORs). κORs in the ventral tegmental area regulate multiple aspects of dopaminergic and non-dopaminergic cell function. Here we show that at GABAergic synapses on rat VTA dopamine neurons, a single exposure to a brief cold-water swim stress induces prolonged activation of κORs. This is mediated by activation of the receptor during the stressor followed by a persistent, ligand-independent constitutive activation of the κOR itself. This lasting change in function is not seen at κORs at neighboring excitatory synapses, suggesting distinct time courses and mechanisms of regulation of different subsets of κORs. We also provide evidence that constitutive activity of κORs governs the prolonged reinstatement to cocaine-seeking observed after cold water swim stress. Together, our studies indicate that stress-induced constitutive activation is a novel mechanism of κOR regulation that plays a critical role in reinstatement of drug seeking.
Keywords: GABA; Kappa Opioid Receptor; Stress; Synaptic Plasticity; VTA; dopamine; neuroscience; rat.
Conflict of interest statement
The authors declare that no competing interests exist.
Figures




References
-
- Becker JA, Wallace A, Garzon A, Ingallinella P, Bianchi E, Cortese R, Simonin F, Kieffer BL, Pessi A. Ligands for kappa-opioid and ORL1 receptors identified from a conformationally constrained peptide combinatorial library. Journal of Biological Chemistry. 1999;274:27513–27522. doi: 10.1074/jbc.274.39.27513. - DOI - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

