Resistance to hypertension mediated by intercalated cells of the collecting duct
- PMID: 28405625
- PMCID: PMC5374064
- DOI: 10.1172/jci.insight.92720
Resistance to hypertension mediated by intercalated cells of the collecting duct
Abstract
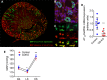
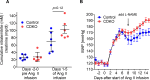
The renal collecting duct (CD), as the terminal segment of the nephron, is responsible for the final adjustments to the amount of sodium excreted in urine. While angiotensin II modulates reabsorptive functions of the CD, the contribution of these actions to physiological homeostasis is not clear. To examine this question, we generated mice with cell-specific deletion of AT1A receptors from the CD. Elimination of AT1A receptors from both principal and intercalated cells (CDKO mice) had no effect on blood pressures at baseline or during successive feeding of low- or high-salt diets. In contrast, the severity of hypertension caused by chronic infusion of angiotensin II was paradoxically exaggerated in CDKO mice compared with controls. In wild-type mice, angiotensin II induced robust expression of cyclooxygenase-2 (COX-2) in renal medulla, primarily localized to intercalated cells. Upregulation of COX-2 was diminished in CDKO mice, resulting in reduced generation of vasodilator prostanoids. This impaired expression of COX-2 has physiological consequences, since administration of a specific COX-2 inhibitor to CDKO and control mice during angiotensin II infusion equalized their blood pressures. Stimulation of COX-2 was also triggered by exposure of isolated preparations of medullary CDs to angiotensin II. Deletion of AT1A receptors from principal cells alone did not affect angiotensin II-dependent COX2 stimulation, implicating intercalated cells as the main source of COX2 in this setting. These findings suggest a novel paracrine role for the intercalated cell to attenuate the severity of hypertension. Strategies for preserving or augmenting this pathway may have value for improving the management of hypertension.
Figures





References
-
- Levine DZ, Iacovitti M, Buckman S, Hincke MT, Luck B, Fryer JN. ANG II-dependent HCO3- reabsorption in surviving rat distal tubules: expression/activation of H(+)-ATPase. Am J Physiol. 1997;272(6 Pt 2):F799–F808. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Research Materials

